Thermal conductivity of gaseous chlorine, use Eq. 9.3-15 to calculate the thermal conductivity of gaseous chlorine. To
Question:
Thermal conductivity of gaseous chlorine, use Eq. 9.3-15 to calculate the thermal conductivity of gaseous chlorine. To do this you will need to use Eq. 1.4-14 to estimate the viscosity, and will also need the following values of the heat capacity:
T (K) 200 300 400 500 600
Cp (cal/g-mole ∙ K) (8.06) 8.12 8.44 8.62 6.74
Check to see how well the calculated values agree with the following experimental thermal conductivity data5
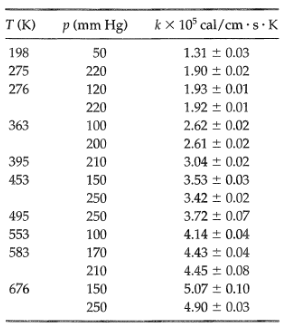
Transcribed Image Text:
kx 10° cal/cm · s·K T (K) p (mm Hg) 198 1.31 + 0.03 50 275 220 1.90 ± 0.02 276 120 1.93 ± 0.01 220 1.92 ± 0.01 363 100 2.62 + 0.02 200 2.61 ± 0.02 395 3.04 + 0.02 210 453 150 3.53 ± 0.03 250 3.42 + 0.02 3.72 ± 0.07 495 250 4.14 + 0.04 553 100 583 170 4.43 + 0.04 210 4.45 + 0.08 5.07 + 0.10 676 150 250 4.90 ± 0.03
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Thermal conductivity of gaseous chlorine For Cl2 Table E1 gives M 1414 then gives 7091 o VMT 2...View the full answer

Answered By

Shridhar Kamble
I have completed my master's degree in physics,with specialization in Space science from a reputed university. I have worked with my juniors on two major projects involving IRNSS/GPS communication and it's applications in probing earth's ionosphere. During these projects I guided my juniors towards understanding the intricacies of trans-ionospheric communications. I have given several seminars and lectures throughout my academic life. These lectures and seminars always recieved a positive response. I have been teaching physics in the local highchool for a few months and I'm looking forward to a promising career in academia
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Q1. approving paper" job. For this you will need a stopwatch, 6 sheets of A4 or A5 paper, pencil case and 3 different colored pencils, a time study form. The definition of this work is that 6 papers...
-
Estimate the cp of nitrogen at 300 kPa and 400 K, using (a) The relation in the above problem and (b) Its definition. Compare your results to the value listed in Table A2b.
-
Estimate the maximum and minimum thermal conductivity values for a cermet that contains 85 vol% titanium carbide (TiC) particles in a cobalt matrix. Assume thermal conductivities of 27 and 69 W/m-K...
-
The unadjusted trial balance of Simple Consulting Services as at December 31, 2021 is as follows: Cash Accounts receivable Prepaid insurance Supplies inventory Office equipment Accumulated...
-
Your company, Rasmussen Consulting, was hired by MovieFlix, a company that provides subscription service for on-demand Internet streaming media and DVD-by-mail within the U.S. You are the consultant...
-
In recent years, a growing array of entertainment options competes for consumer time. By 2004, cable television and radio surpassed broadcast television, recorded music, and the daily newspaper to...
-
Fill in the Blank. Vibration neutralizer is also known as dynamic vibration ____________.
-
Gale Brewer, CPA, has been the partner in charge of the audit of Merkle Manufacturing Company, a nonpublic company, for 13 years. Merkle has had excellent growth and profits in the past decade,...
-
1. A circular disk of radius 3.0 cm has a uniform surface charge density of 47 nC/m'. What is the total charge on the disk? 2. A sphere of radius 2.4 cm has a uniform volume charge density of 354...
-
Cajun Company has outstanding 2,500 shares of $100 par, 6% preferred stock and 15,000 shares of $10 par value common. The following schedule shows the amount of dividends paid out over the last 4...
-
Thermal conductivity and Prandtl number of a polyatomic gas. (a) Estimate the thermal conductivity of CH 4 at 1500K and 1.37 atm. The molar heat capacity at constant pressure 4 at 1500K is 20.71...
-
Thermal conductivity of chlorine-air mixtures, Using Eq. 9.3-17, predict thermal conductivities of chlorine-air mixtures at 297K and 1 arm for the following mole fractions of chlorine: 0.25, 0.50,...
-
Modify Prob. 2.146 as follows: Let the 10-cm-diameter sphere be concrete (SG = 2.4) hanging by a string from the top. If the tank accelerates to the right at 5 m/s/s, at what angle will the balloon...
-
ichier Answer 10 of the 11 following questions. You may omit one of your choosing. All questions count equally though all are not equally difficult. 1. Find the general antiderivative of =sin(x (x) +...
-
Section D: Relative Risk The Relative Risk measures the strength of the association that a risk factor or exposure has with an outcome. It is interpreted based on 1 representing no association. A...
-
A producer of printed circuit boards (PCBs) wishes to estimate the scoring depth of their circuits. To this end, they have collected 45 samples of measured depths. The mean and standard deviations of...
-
Yeats, Irons, and Rhoades (1975) found that annual deposit growth for 48 commercial banks can be modeled by the following function: D_(t+1)/D_t=1.172-0.125t^-1+1.135t^-2 where D is the year-end...
-
Match the histograms on the right to the summary statistics given Standard Mean Median Deviation 1 52 52 15 60 60 9 52 52 10 IV 52 52 18 (a) (b) (c) T (d) Frequency fouenbai Frequency 10- 10- 30 (b)...
-
Deja runs her own hot dog stand on the U of A campus. The monthly cost of the cart rental and business permit is \($300\). Dejas contribution margin per unit is \($1.50\) and her contribution margin...
-
What impact has the Internet had on the globalization of small firms? How do you think small companies will use the Internet for business in the future?
-
Assuming the volumes are additive, what is the [Cl - ] in a solution obtained by mixing 225 mL of 0.625 M KCl and 615 mL of 0.385 M MgCl 2 ?
-
Most students borrow to finance their college educations and repay the loans later. How might the life-cycle hypothesis help to explain this behavior?
-
Late 2010, Congress renewed for a two-year period the tax cuts first passed by the Bush administration. The original Bush tax cuts were set to expire in 2011, 10 years after their original passage....
-
What are the three categories of consumption? Which category is the most volatile? Which category is the least volatile?
-
The figure below shows forces acting at various points on a metal shaft. The angles a = 40, = 29, y = 17. The length = 4.4 m. Find the net torque (in N m) on the shaft about the following axes. 25 N...
-
How can cross-functional teams, comprising members from diverse departments and disciplines, navigate the challenges of differing perspectives, knowledge bases, and work processes to achieve project...
-
What are the unique challenges and opportunities associated with virtual teams and remote collaboration, and how can technology, communication tools, and best practices be leveraged to overcome...

Study smarter with the SolutionInn App


