This alkynes hydration reaction can occur without added Hg2+. Show all the steps in themechanism. H,SO, PhC=CH
Question:
This alkynes hydration reaction can occur without added Hg2+. Show all the steps in themechanism.
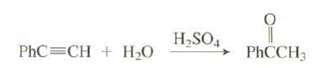
Transcribed Image Text:
H,SO, PhC=CH + H20 PHCCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
PhCCH PhCCH 0 H 1 ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show all the steps in the mechanisms for these reactions. Don't forgot to use curved arrows to show the movement of electrons in eachstep. CH3 CH3 ) . + CHH ., + Br CH; CH3 H,SO, CH;CH,OCH,CH; + H;O...
-
Show all the steps in the mechanism for these reactions. Include stereochemistry where it is important.
-
Show all the steps in the acid-catalyzed formation of a cyclic acetal from ethylene glycol and an aldehyde or ketone.
-
Question 4: What is meant by statistical versus nonstatistical and probabilistic versus nonprobabilistic sample selection?
-
Who is Edward Tufte? Why do you think we should know about his work?
-
Using the rules of contours, list the contouring mistakes that are shown in the accompanying figure and list the contouring rule it violates. 68.96 Problem 17.21
-
CD Real Estate specializes in residential real estate services in the state of California. To complement the experience and local market knowledge of its licensed realtors, CD Real Estate wants to...
-
The diagram below displays short-run cost curves for a facility that produces liquid crystal display (LCD) screens for cell-phones: a. What are the daily total fixed costs of producing LCD screens?...
-
If a bank charges an interest rate of 0.6% per quarter on loans to its customers, what APR must this lender report to consumers? A bond with 10 years left to maturity currently sells for 105% of par...
-
Your client Kevins Train Spotting Limited holds a 25-year lease dated 01/01/2000 with 5 yearly reviews. The rent passing is currently 92,000 per annum. The Companys Director has approached you with a...
-
Show syntheses of these compounds from the indicated starting materials. More than one step may b necessary. Your syntheses may produce both enantiomers of any target that ischiral. , CH,CH,CH3 a)...
-
Explain which compound has a faster rate of reaction withHCI: b) or or NO2 or
-
The thin-wall tank is fabricated from a hemisphere and cylindrical shell. Determine the vertical reactions that each of the four symmetrically placed legs exerts on the floor if the tank contains...
-
How much force is being experienced by each of the lifter's hands if the bar has a mass of 164 kg and she has a mass of 85 kg. You may use 10 m/s2 for gravity, leave the answer to zero decimal places...
-
As loan analyst for Culver Bank, you have been presented the following information. Toulouse Co. Lautrec Co. Assets Cash $115,000 $316,000 Receivables 211,000 305,000 Inventories 595,000 539,000...
-
During the last five years there has been an inflationary trend in the average cost of a gallon of milk, as the following chart shows. Calculate the y-intercept of the regression line. (show your...
-
Price and cost (dollars per client) 100.00 90.00 80.00 70.00 60.00 50.00 40.00 30.00 20.00 10.00 0 MR 6 MC ATC D 8 10 Quantity (clients per day) 10. Kevin owns a personal training gymnasium in...
-
How can a college basketball coach use lean system strategies to improve the team's performance and win the national championship? What wastes can be eliminated from the team's training and...
-
Impact of September 11 on Air Travel in the United States. The Research and Innovative Technology Administrations Bureau of Transportation Statistics conducted a study to evaluate the impact of the...
-
A sprinkler head malfunctions at midfield in an NFL football field. The puddle of water forms a circular pattern around the sprinkler head with a radius in yards that grows as a function of time, in...
-
Which of the following conditions indicate an acidic solution at 25C? a. pH = 3.04 b. [H]> 1.0 10-7 M c. pOH = 4.51 d. [OH-] 3.21 10- M
-
What is the stereo chemical relationship of D-ribose to L-xylose? What generalizations can you make about the following properties of the two sugars? (a) Melting point (b) Solubility in water (c)...
-
All aldoses exhibit mutarotation. For example, -D-galactopyranose has []D = + 150.7, and -D-galactopyranose has []D = + 52.8. If either anomer is dissolved in water and allowed to reach equilibrium,...
-
How many D-2-ketohexoses are possible? Draw them.
-
Explain possible advantages and disadvantages of PSL's current international trading strategy (subsidiary in China, export & import and online presence).
-
In November 2020 you entered into four May 2021 long futures contracts for crude oil (1,000 barrels per contract) for $65.00 per barrel. If the futures price was $70.00 per barrel on December 31,...
-
4) Champagne, Inc., had revenues of $13 million, cash operating expenses of $9 million, and depreciation and amortization of $2 million during 2008. The firm purchased $650,000 of equipment during...

Study smarter with the SolutionInn App


