Three different liquids are used in the manometer shown here. (a) Derive an expression for p 1
Question:
Three different liquids are used in the manometer shown here.
(a) Derive an expression for p1 ? p2 in terms of pA, pB, pC, h1, and h2.
(b) Suppose fluid A is methanol, B is water, and C is a manometer fluid with a specific gravity of 1.37; Pressure p2 = 121.0kPa; h1 = 30.0cm; and h2 = 24.0cm. Calculate p1 (kPa)
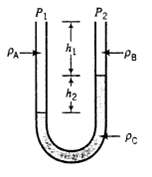
PA Pi 下 + P₂ 阳 Po
Step by Step Answer:
a Ppgh P Pgh Pcgh RP PBPgh PcPgh b ...View the full answer



Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Related Video
In Physics, a force is an influence that can change the motion of an object. A force can cause an object with mass to change its velocity, i.e., to accelerate. Force can also be described intuitively as a push or a pull. A force has both magnitude and direction, making it a vector quantity. In this video, the concept of force and torque is demonstrated with the practical application of changing the flat tire of the car.
Students also viewed these Chemical Engineering questions
-
With reference to Fig. 5-10, (a) Derive an expression for the vector magnetic potential A at a point P located at a distance r from the wire in the xy plane, and then (b) Derive B from A. Show that...
-
Derive an expression for the heat flux per unit area at depth x and time when a semi-infinite solid is suddenly exposed to an instantaneous energy pulse at the surface of strength Q0/A.
-
Derive an expression for the capillary height change h, as shown, for a fluid of surface tension Y and contact angle θ between two parallel plates W apart. Evaluate h for water at 20°C if W...
-
SG Company acquired 80% of Popsters Company on January 1, 2019, when the stockholders equity of Popsters consisted of: Ordinary shares, P100 par P500,000 Paid in capital in excess of par 400,000...
-
What are the problems in measuring the unemployment rate? In what ways does the official BLS measure of the unemployment rate understate the true degree of unemployment? In what ways does the...
-
(a) How do you recognize the independent variable of an experiment? (b) How do you recognize the dependent variable?
-
How has religious practice changed in the United States?
-
Stiever Company estimates that variable costs will be 60% of sales, and fixed costs will total $800,000. The selling price of the product is $4. Instructions (a) Prepare a CVP graph, assuming maximum...
-
Determine the yield-to-call (to nearest 0.1 of a percent) of anLTV bond with a 14 percent coupon, that pays interest semiannually.The bond can be called in 7 years, has a call premium of $140, andis...
-
Import Distributors, Inc. (IDI), imported appliances and distributed them to retail appliance stores in the Rocky Mountain states. IDI carried three broad lines of merchandise: audio equipment...
-
Two mercury manometers, one open-end and the other sealed-end, are attached to an air duct. The reading on the open-end manometer is 25 mm and that on the sealed-end manometer is 800mm. Determine the...
-
The level of toluene (a flammable hydrocarbon) in a storage tank may fluctuate between 10 and 400 cm from the top of the tank. Since it is impossible to see inside the tank, and open-end manometer...
-
Let a 0. Solve |x| = 3.
-
Analyze Telstra's Twitter account using Leximancer and provide an overview of the result. please provide a picture of them (the result you get on the Leximancer website) as you analyze Telstra's...
-
K Harry Taylor plans to pay an ordinary annuity of $5,200 annually for ten years so he can take a year's sabbatical to study for a master's degree in business. The annual rate of interest is 2.5%....
-
1. Perform a sensitivity analysis on each of the key variables listed below, assuming they can each vary from their base-case or expected value by 10 percent. unit sales growth rate in unit sales...
-
Case 1: Use AFN Formula and calculate additional funds needed for Year 20x4E. Assume that MMI Company's total revenue will grow at 30% next year, and currently a company is fully utilized its fixed...
-
Refer to Table 10-4. a. What was the settlement price on the June 2020 5-Year U.S. Treasury Notes futures contract on March 13, 2020? (Do not round your intermediate calculations. Round your...
-
A 1.6-m uniform rod is being used to balance two buckets of paint, each of inertia \(m\), one at each end of the rod. (a) Where is the pivot located? (b) If paint is removed from one bucket until its...
-
In a system with light damping (c < cc), the period of vibration is commonly defined as the time interval d = 2/d corresponding to two successive points where the displacement-time curve touches one...
-
How are total revenue and price elasticity of demand related?
-
You have analyzed an aqueous ammonia solution and find that it contains 30 wt% NH3. (a) Use Figure 8.5-2 to determine the mass fraction of NH3 in the vapor that would be in equilibrium with this...
-
An NH3 H2O mixture containing 60 wt% NH3 is brought to equilibrium in a closed container at 140F. The total mass of the mixture is 250 g. Use Figure 8.5-2 to determine the masses of ammonia and of...
-
An ammonia solution at a high pressure is flash-vaporized at a rate of 200lbm/h. The solution contains 0.70lbm NH3/lbm, and its enthalpy relative to H2O (1, 32F) and NH3 (1, 40F) is 50 Btu/lbm....
-
Diverse Perspectives on the World Economy assignment You are required to consider a contemporary economic or social issue that is currently attracting media attention. It could be a local, national,...
-
Daniel made the following transactions during the first week of January. 1 January Buys stock for 350 on credit 3 January Makes sales of 740 on credit 4 January Cash paid to a supplier of 120, for...
-
Please answer all parts of the following question. Given the following information: Total Product (TP) Price Fixed Cost (TFC) Variable Cost (TVC) 0 $1000 $600 $ 0 1 1000 600 1200 2 1000 600 2000 3...

Study smarter with the SolutionInn App


