Three hundred L/h of a 20 mole% C 3 H 8 ?80% n-C 4 H 10 gas
Question:
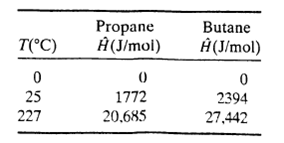
Three hundred L/h of a 20 mole% C3H8?80% n-C4H10 gas mixture at 0oC and 1.1 atm and 200 L/h of a 40 mole% C3H8?60% n-C4H10 mixture at 25?C and 1.1 atm are mixed and heated to 227?C at constant pressure. Calculate the heat requirement in kJ/h. Enthalpies of propane and n-butane are listed below. Assume ideal gas behavior.
Transcribed Image Text:
T(°C) 0 25 227 Propane Ĥ (J/mol) 0 1772 20,685 Butane Ĥ(J/mol) 0 2394 27,442
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Basis Given feed rates n molh 02 C3Hg 08 C4H10 0C 11 atm Q kJh Mol...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Question Posted:
Students also viewed these Chemical Engineering questions
-
Propane gas is burned steadily at 1 atm pressure with a 10 percent excess oxygen supplied by atmospheric air. The reactants enter a steady flow combustor at 25C. Determine the final temperature of...
-
Butane gas at 77 F is mixed with 150% theoretical air at 1000 R and is burned in an adiabatic SSSF combustor. What is the temperature of the products exiting the combustor?
-
In Pissarides's view of labor markets (job matching model), the focus is on finding a good ________. a. level of TFP (A) b. supply of money (M). c. match d. Wage
-
Why is it reasonable to assume that most firms will have a banking relationship?
-
During the last few years, Jana Industries has been too constrained by the high cost of capital to make many capital investments. Recently, though, capital costs have been declining, and the company...
-
The probability that the noise level of a wide-band amplifier will exceed \(2 \mathrm{~dB}\) is 0.05. Use Table 1 or software to find the probabilities that among 12 such amplifiers the noise level...
-
You are one of three carpenters assigned to complete a short construction project. Right before the start of the project, one of your fellow carpenters was hospitalized and will not be available to...
-
2. (3 points) Herron Company operates its business solely in Jurisdiction H, which levies a 5.0% sales and use tax. This year, Herron paid $1,270,000 to purchase tangible property from a dealer...
-
1.3 Determine the force P necessary for the equilibrium of a steel bar shown in Fig. 1.69. The diameters of the first, middle and the last segments of the bar are 30 mm, 25 mm and 30 mm respectively....
-
The following diagram shows a simplified version of how a refrigerator works: In a liquid receiver (1), a liquid refrigerant (any one of a number of halogenated hydrocarbons such as CC1 2 F 2 ) is...
-
Air at 38C and 97% relative humidity is to be cooled to 18C and fed into a plant area at a race of 510m3/min. (a) Calculate the rate (kg/mm) at which water condenses. (b) Calculate the cooling...
-
What are the advantages of user-defined enumeration types?
-
How is Earths lithosphere like a jigsaw puzzle? How is it NOT like a jigsaw puzzle?
-
In what way is Earth like a jar of water and oil left standing?
-
Smash a few salt cubes and then look at them carefully again. What youll see are smaller salt cubes. Explain why salt breaks into cubes in terms of the property of cleavage.
-
Why does folding occur deep below Earths surface?
-
Why are the ferromagnesian silicates often dark, dense, and magnetic?
-
Suppose that the fine for illegally parking a car is \( 3,200\) and that a fine amount of 1,760 is sufficient for discouraging most people from parking their cars illegally. How high must the...
-
Discuss the information available from the following techniques in the analysis of inorganic pigments used in antique oil paintings: (i) Powder X-ray diffraction, (ii) Infrared and Raman...
-
Solve the differential equation. y" + 4y' + 4y = 0
-
Calculate the molar H3O+ ion concentration of a solution that has a pH of (a) 4. 31. (b) 0.59. (c) 7.62. (d) 20.76.
-
Calculate the p-functions for each ion in a solution that is (a) 0.0300 M in NaBr. (b) 5.5 3 10-3 M in Ba(OH)2. (c) 8.7 3 10-3 M in CaCl2 and 6.6 x 10-3 M in BaCl2
-
Convert the following p-functions to molar concentrations: (a) pH = 1.020. (b) pBr = 7.77. (c) pLi = 12.35. (d) pMn = 0.135.
-
1 0 . What type of passing parameter based on the following code fragment? class Student { public static void main ( String [ ] args ) { Student s = new Student ( Ali , 2 1 ) ; printData ( s ) ; }...
-
Observe changes occurred in value of all registers, which is accessed by operand in Debug Mode, then fill the blanks. (Write all esi value in L1 and ebx value in L2) TITLE Practice08-1 INCLUDE...
-
ALGORITHM Brute ForceClosest Pair(P) //Finds distance between two closest points in the plane by brute force //Input: A list P of n (n 2) points p(x1, y),..., Pn(xn, yn) //Output: The distance...

Study smarter with the SolutionInn App


