Treatment of 1, 1-diphenyl- 1, 2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a
Question:
Treatment of 1, 1-diphenyl- 1, 2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction.
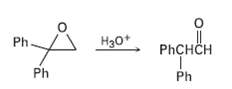
Transcribed Image Text:
Ph H30+ PHCHCH Ph Ph
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
HOH PhCCH PhCCH OH PhCCH HO PhCHCH H h...View the full answer

Answered By

Rehab Rahim
I am well versed in communicating and teaching in areas of all business subjects. I have helped many students in different ways from answering answers to writing their academic papers.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanism begins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilic acyl substitution...
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Propose a mechanism for the reaction of (a) 1-methylcyclohexanol with HBr to form 1-bromo-1-methylcyclohexane. (b) 2-cyclohexylethanol with HBr to form 1-bromo-2-cyclohexylethane?
-
U Ltd has three subsidiaries (one of which is dormant) and has the following results for the year to 31 December 2020: Compute the corporation tax liability for the year and state the date (or dates)...
-
Describe the types of information employees at an Apple store require and compare it to the types of information the executives at Apple's corporate headquarters require. Are there any links between...
-
Find examples of an offshore factory, source factory, server factory, contributor factory, outpost factory, and lead factory.
-
Identify the departments reporting to the vice-president of production?
-
Effects of the level of production on an outsourcing decision Seymour Chemical Company makes a variety of cosmetic products, one of which is a skin cream designed to reduce the signs of aging....
-
Flight Caf Planning Budget For the Month Ended July 31 27,000 $ 108,000 Budgeted meals (q) Revenue ($4.00q) Expenses: Raw materials ($1.80q) Wages and salaries ($6,100 + $0.20q) Utilities ($2,000 +...
-
Walkers is planning to acquire Cayman Bank, a freestanding C corporation, in expectation that new management can be brought in to achieve substantial operating efficiencies. You have been retained to...
-
How would you synthesize racemic disparlure (Problem 18.46) from corn- pounds having ten or fewer carbons?
-
How would you prepare o-hydroxyphenyl-acetaldehyde from phenol? More than one step is required. HO o-Hydroxyphenylacetaldehyde CH-CO
-
Briefly explain the suggestions that consumers should follow when shopping for a homeowners policy.
-
Given the following information, calculate each organization's times-interest-earned ratio and debt ratio. Which organization is in the stronger position? Seven Kingdoms Corp Snow Corp. Net Income...
-
5. Unilever's cost of goods sold for 2014 is $36 million. How much inventory (in $) does the company hold if it has a one-month supply of inventory on average
-
6. Why is a color-blind ideology problematic? Why can't we ignore "color" and create a raceless society? 7. How does Whiteness function as a location of structural advantage? What is your...
-
1. In each of the following cases, draw a graph showing an increase or decrease in equilibrium interest rates. a. Suppose the demand for loanable funds remains unchanged. Will the equilibrium...
-
In 1999, GLSEN identified that little was known about the school experiences of lesbian, gay, bisexual, transgender, and queer (LGBTQ+) youth and that LGBTQ+ youth were nearly absent from national...
-
Ong Enterprises had the following selected transactions. 1. Shareholders invested NT\($40,000\) cash in the business in exchange for ordinary shares. 2. Paid office rent of NT\($8,400. 3. Performed...
-
Swifty company is a publicly held corporation whose $1 par value stock is actively traded at $30 per share. The company issued 3400 shares of stock to acquire land recently advertised at $93000. When...
-
In a foundry, metal castings are cooled by quenching in an oil bath. Typically, a casting weighing 20 kg and at a temperature of 450 C is cooled by placing it in a 150-kg involatile oil bath...
-
Use arguments based on resonance and electronegativity effects to explain the trend in carbonyl IR stretching frequencies from higher frequency for esters and carboxylic acids to lower frequencies...
-
Classify each of the following compounds as an alkane, alkene, alkyne, alcohol, aldehyde, amine, and so forth. (a) (b) CH3-C¡CH (c) (d) (e) (f) Obtained from oil of cloves -2. Sex attractant...
-
Boron trifluoride (BF3) has no dipole moment ( = 0 D). Explain how this observation confirms the geometry of BF3 predicted by VSEPR theory.
-
Create a segmentation, selection and focus model that allows the PUMA company in Mexico to determine its customer, where to find it and how to generate value given its model. 7) Selection of the most...
-
Determine if the following values are zeroes polynomial. 4 3 f(x) = x + x-18x - 16x + 32 x+1, +2, +4 -
-
Cali Windows is a small company that installs windows. Its cost structure is as follows: (Click the icon to view the cost structure.) Calculate (a) the breakeven point in units and revenues and (b)...

Study smarter with the SolutionInn App


