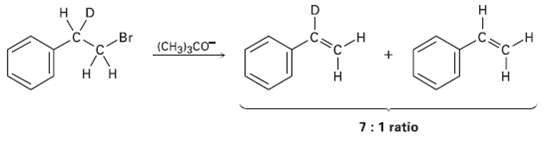
Treatment of 1 .bromo-2-deuterio-2-phenylethane with strong base leads to a mixture of deuterated and non-deuterated phenylethylenes in
Question:
Treatment of 1 .bromo-2-deuterio-2-phenylethane with strong base leads to a mixture of deuterated and non-deuterated phenylethylenes in an approximately 7: 1 ratio.Explain.
Transcribed Image Text:
н Br (CH3)3CO 7:1 ratio O-エ エー ローU
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (11 reviews)
Since carbondeuterium bonds are slightly stronger than carb...View the full answer

Answered By

Akshay Singla
as a qualified engineering expert i am able to offer you my extensive knowledge with real solutions in regards to planning and practices in this field. i am able to assist you from the beginning of your projects, quizzes, exams, reports, etc. i provide detailed and accurate solutions.
i have solved many difficult problems and their results are extremely good and satisfactory.
i am an expert who can provide assistance in task of all topics from basic level to advance research level. i am working as a part time lecturer at university level in renowned institute. i usually design the coursework in my specified topics. i have an experience of more than 5 years in research.
i have been awarded with the state awards in doing research in the fields of science and technology.
recently i have built the prototype of a plane which is carefully made after analyzing all the laws and principles involved in flying and its function.
1. bachelor of technology in mechanical engineering from indian institute of technology (iit)
2. award of excellence in completing course in autocad, engineering drawing, report writing, etc
4.70+
48+ Reviews
56+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Treatment of 3, 4-dibromohexane with strong base leads to loss of 2 equivalents of HBr and formation of a product with formula C6H10. Three products are possible. Name each of the three, and tell how...
-
Aldol condensation of 3-methylcyclohexanone leads to a mixture of two enone products, not counting double-bond isomers draw them.
-
A container holds a mixture of three non reacting gases: 2.40 mol of gas 1 with Cv1 = 12.0 J/mol ?? K, 1.50 mol of gas 2 with Cv2 = 12.8 J/mol ?? K and 3.20 mol of gas 3 with Cv3 = 20.0 J/mol ?? K....
-
Modesto Trading Berhad has recently appointed Messrs. Suresh Kumar & Co., Chartered Accountants, to audit the companys financial statements for the year ended 30 June 2021. You are assigned by...
-
Do you think an outside contractor could provide training for Nick's as effectively as its current methods do? Why or why not? Are there some types or topics of training for which a contractor might...
-
Using the results in column (7): a. Akira is a man with 10 years of driving experience. What is the probability that he will pass the test? b. Jane is a woman with 2 years of driving experience. What...
-
The following data come from a study in which random samples of the employees of three government agencies were asked questions about their pension plan: Use the 0.01 level of significance to test...
-
Consider Boeing (a producer of jet aircraft), General Mills (a producer of breakfast cereals), and Wacky Jacks (which claims to be the largest U. S. provider of singing telegrams). For which of these...
-
A program has created an Object called "friend." This object includes the following fields: firstName, bDay, favColour and phoneNumber. An ArrayList of the object friend is declared with the...
-
Gold Sporting Equipment (GSE) is in the process of preparing its budget for the third quarter of 2010. The budgeting staff has gathered the following data: 1. Account balances as of June 30: 3. Sales...
-
Reaction of HBr with (R)-3-methyl-3-hexanol leads to racemic 3-brorno-3-mcthylhcxane.Explain. OH CH3CH2CH2CCH2CH3 3-Methyl-3-hexanol H3
-
A Propose a structure for an alkyl halide that gives only (E)-3-methyl-2-phenyl-2-pentene on E2 elimination, Make sure you indicate the stereochemistry.
-
The block has a mass of 20 kg and is released from rest when s = 0.5 m. If the mass of the bumpers A and B can be neglected, determine the maximum deformation of each spring due to the collision. s =...
-
Choose two listed companies from Bursa Malaysia, Using their most recent financial reports and stock price performance, compare, decide, and advise which company has the greatest potential for growth...
-
A residential multi-story building is planned to be constructed on a reinforced concrete raft resting on the soil formation shown in the figure below. This requires excavation of 5 feet below...
-
ASSA ABLOY's debt situation was good. Their debt decreased from 2018 to 2020 but increased significantly in the years following. According to Buffett, ASSA ABLOY is not a durable, competitive company...
-
(b) (c) Let A = {0,1,2). Find (4 - {(0,0))) A. Let f: R R and g: RR, R is the set of real numbers. Find f g and gof, where f(x) = x - 2 and g(x)= x + 4. State whether these functions are injective,...
-
You are trying to decide between investing in a TFSA or an RRSP . Describe the two different investments ( contribution amount, liquidity, tax implications etc.) and explain which one you would most...
-
A Payne County commissioner has \($20\),000 remaining in the budget to spend on one of three worthy projects. Each is a one-time investment, and there would be no follow-on investment, regardless of...
-
Revol Industries manufactures plastic bottles for the food industry. On average, Revol pays $76 per ton for its plastics. Revol's waste-disposal company has increased its waste-disposal charge to $57...
-
What is the effect of a change in concentration of a reactant or product on a chemical reaction initially at equilibrium?
-
(a) Explain why the following substituted acetic acid cannot be formed by the malonic ester synthesis. (b) Sections 22-2B and 22-3 showed the use of lithium diisopropylamide (LDA) to deprotonate a...
-
Show the ketones that would result from hydrolysis and decarboxylation of the following β-keto esters. (a) (b) (c) PhCH,-CH-C-CH COOC,H C-C-CH COOCH,CH OCH,CH3
-
Predict the major products of the following reactions. (a) (b) (c) (1) LDA (2) CH2=CHCH-Br acetone LDA (2) CH,CHI (1) LDA ()CHI
-
2. What basic criteria would you recommend that K&G set to determine what jobs the company should accept in the future?
-
Why are Incoterms 2020 and Terms of Payment considered an important factor in executing global trade contracts? and also provide five various examples and risk related risk factors,
-
What is the purpose of using the Loan Manager in QuickBooks?

Study smarter with the SolutionInn App


