Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction. For example,
Question:
Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction. For example, treatment of Cyclohexanone with diazomethane yields Cycloheptanone. Propose amechanism.
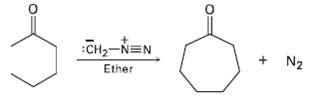
Transcribed Image Text:
Сна-NEN Ether N2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
CH NEN 0 CHNEN 18 ...View the full answer

Answered By

Douglas Makokha
Unlock Academic Success with Dedicated Tutoring and Expert Writing Support!
Are you ready to excel in your academics? Look no further! As a passionate tutor, I believe that dedication and hard work are the keys to achieving outstanding results. When it comes to academics, I strive to provide nothing but the best for every student I encounter.
With a relentless thirst for knowledge, I have extensively researched numerous subjects and topics, equipping myself with a treasure trove of answers to tackle any question that comes my way. With four years of invaluable experience, I have mastered the art of unraveling even the most intricate problems. Collaborating with esteemed writers has granted me exclusive access to the trade secrets utilized by the industry's top professionals.
Allow me the pleasure of assisting you with your writing assignments. I thrive on challenges and will guide you through any obstacles you may face. Together, we will unlock your academic potential and pave the way for your success.
4.90+
60+ Reviews
345+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the acid-catalyzed reaction of cyclohexanone with pyrrolidine.
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
A standard synthetic sequence for building a six-membered cyclic ketone onto an existing aromatic ring is shown in outline as follows. Specify the reagents necessary for each step. CCH2CH2COH CH...
-
Governmental Funds Statement of Revenues Expenditures and Changes in Fund Balance. You have recently started working as the controller for a small county. The county is preparing its financial...
-
How can you determine when it is appropriate to send an e-mail, a memo, or a letter?
-
If A and B are events, then P(A|B) P(B). In Problem, discuss the validity of each statement. If the statement is always true, explain why. If not, give a counterexample.
-
Explain the concept of proof by refutation.
-
Refer to Exhibit 8-14 regarding Greyson Technology??s launch of a new digital communications device. Suppose that Greyson reduced the quarterly spending on product development in panel A, which...
-
Consider the LC circuit shown below. Switch S is initially open, and the capaci- tor has a charge Qm on its plates. At t = 0 the switch is closed. C L -0000 Qm S At time t = LC, what is the ratio of...
-
On January 1, 2019, Perini Company purchased an 85% interest in Silvas Company for $400,000. On this date, Silvas Company had common stock of $90,000 and retained earnings of $210,000. An examination...
-
The Favorskii reaction involves treatment of an ?-bromo ketone with base to yield a ring-contracted product. For example, reaction of 2-bromocyclo-hexanone with aqueous NaOH yields...
-
Ketones react slowly with benzeneselenenyl chloride in the presence of HCl to yield ?-phenylselcno ketones. Propose a mechanism for this acid-catalyzed a-substitution reaction. CeHgSeCi Se-C6H5
-
Which of the following expenses would you regard as part of the cost of goods sold? (a) Advertising the goods for sale (b) Insurance and freight on importing goods for sale (c) Packing the goods for...
-
Anderson Corporation just paid a dividend of $3.65 per share. The company will increase its dividend by 20% next year and will then decrease its dividend growth rate by 5% per year until it reaches...
-
What is the current yield of a bond with a 8% coupon rate coupon paying interest annually, 5 years until maturity, a face value of $1,000, and a current price of $971?
-
If federal court judges interpret the Constitution according to its historical creation, and based on the original intent of the Founding Fathers of the U.S. Constitution, then this is which type of...
-
A bond pays interest semi-annually and sells for $1100. It has six years left to maturity and a par value of $1,000. What is its coupon if its promised YTM is 13%?
-
Current Attempt in Progress Tamarisk Company traded a used welding machine (cost $11,880, accumulated depreciation $3,960) for office equipment with an estimated fair value of $6,600. Tamarisk also...
-
Rain falls on land. Then what happens to it?
-
Using thermodynamic data from Appendix 4, calculate G at 258C for the process: 2SO 2 (g) + O 2 (g) 88n 2SO 3 (g) where all gases are at 1.00 atm pressure. Also calculate DG8 at 258C for this same...
-
Determine the molecular geometry of CBr 4 . a) Linear b) Trigonal planar c) Tetrahedral d) Trigonal pyramidal
-
Explain why the differences between the first and second pKa values of the dicarboxylic acids become smaller as the lengths of their carbon chains increase (Table 20.3). TABLE 20.3 pK, Values of Some...
-
What is the pH of a solution containing a buffer consisting of acetic acid and sodium acetate in which the actual [acetic acid]/[sodium acetate] ratio is (a) 1/3? (b) 3? (c) 1?
-
Show why HBr is a stronger acid in acetic acid solvent than it is in water.
-
Solve the initial value problem. dy =3e3t sin (e3t-27), y(In 3)=0 dt The solution is y =
-
Solve by factoring. 5x2-16x+12=0
-
Find f-1 (x) for f (x) = 15 + 12x.

Study smarter with the SolutionInn App


