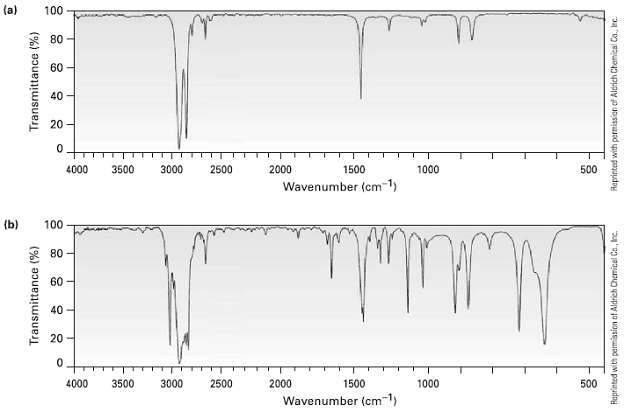
Two infrared spectra are shown. One is the spectrum of cyclohexane, and the other is the spectrum
Question:
Two infrared spectra are shown. One is the spectrum of cyclohexane, and the other is the spectrum of cyclohexane. Identify them, and explain youranswer.
Transcribed Image Text:
(a) 100 80 60 20 - 1000 4000 3500 3000 2500 2000 1500 500 Wavenumber (cm-1) (b) 100 80 60 40 20 - 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm1) Transmittance (%) Transmittance (%) 40 Raprinted with permission of Aldrich Chemicel Co., Inc. Reprinted with permission of Aldrich Chamical Ca. lec.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
Spectrum b differs from spectrum a in several respects Note in part...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Infrared spectra are used by chemists to help identify an unknown substance. Atoms in a molecule that are bound together by a particular bond vibrate at a predictable frequency, and light at that...
-
The infrared spectra for three compounds are provided. Each compound has one or more of the following functional groups: conjugated ketone, ester, amide, nitrile, and alkyne. Determine the functional...
-
Two mass spectra are shown in figure. One spectrum is that of 2-methyl-2-pentene; the other is of 2-hexene. Which is whichexplain? (a) 60 40 20 0. 20 60 80 120 140 10 40 100 m/z (b) 100 80 60 - 10 20...
-
Carey Company is borrowing $200,000 for one year at 12 percent from Second Intrastate Bank. The bank requires a 20 percent compensating balance. What is the effective rate of interest? What would the...
-
Explain why prices are usually strategic complements and capacities are usually strategic substitutes.
-
Consider the Lewis structure for glycine, the simplest amino acid: (a) What are the approximate bond angles about each of the two carbon atoms, and what are the hybridizations of the orbitals on each...
-
1. What was the first thing that PM Solutions did when it was hired by VELCO? 2. Why was the VELCO project compared to a political campaign? 3. Why do you think VELCO was so successful in cutting the...
-
The Hardware Warehouse is evaluating the safety stock policy for all its items, as identified by the SKU code. For SKU M4389, the company always orders 80 units each time an order is placed. The...
-
what are potential risks of Juan and Elena Hernandez's disciplinary style on the psychological development of their children. Identify one theoretical perspective that can be used to explain the...
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
How could you use infrared spectroscopy to distinguish between the following pairs of isomers? (a) HC CCH2NH2 and CH3CH2C N (b) CH3COCH3 and CH3CH2CHO
-
At what approximate positions might the following compounds show IRabsorptions? c??H? " alt="At what approximate positions might the following compounds show IRabsorptions? 51524" class="fr-fic...
-
Refer to the IC counter circuit in Figure 7-107(b): (a) Describe the counters output on QD QC QB QA if START is LOW. (b) Describe the counters output on QD QC QB QA if START is momentarily pulsed LOW...
-
Wildhorse Co. took a physical inventory on December 31 and determined that goods costing $198,500 were on hand. Not included in the physical count were $25,000 of goods purchased from Waterway...
-
The American Snow - blower Company ( ASC ) has the option to make or buy a component part for one of its deluxe snow - blower models. The annual requirement is 8 , 2 0 0 units. A supplier is able to...
-
Determine for and fy, if == (A) f(x, y) = (sin()) In(y) f = fy (B) f(x, y) =sin( In(y)) sin(In(y)) fx fy =1
-
5) Simplify or do the operation on the following formulas: 1) 4a 9 2) (2x + y) = 3) (xa)b+ (xb)a= 4) (8x). (2x-2)=
-
Factor: 125x3+512
-
You are performing an experiment similar to the one shown in Figure 10. 1. The ball is dropped from a height of \(y_{i}=0.200 \mathrm{~m}\) on the pole above the cart just as the pole and cart pass...
-
Currently, there are five concepts of food stands, including: hot dogs, soft pretzels, turkey legs, sandwich wraps, and funnel cakes. This approach will double the existing number of food stands...
-
When HNO 2 is dissolved in water, it partially dissociates according to the equation HNO 2 H + + NO2 . A solution is prepared that contains 7.050 g of HNO 2 in 1.000 kg of water. Its freezing point...
-
For each molecular formula, draw all the possible constitutional isomers of alcohols with that formula. Give the IUPAC name for each alcohol. (a) C3H8O (b) C4H10O (c) C3H6O (d) C3H4O
-
Starting from bromobenzene and any other reagents and solvents you need, show how you would synthesize the following compounds. Any of these products may be used as starting materials in subsequent...
-
Give a systematic (IUPAC) name for each alcohol. Classify each as primary, secondary, or tertiary. (a) (b) (c) (d) (e) (f) (g) Br_CHCH3 OH Cl OH Br OH CI CH2OH
-
Allan Bakke, a thirty-five-year-old white man, had twice applied for admission to the University of California Medical School at Davis. He was rejected both times. The school reserved sixteen places...
-
Consider a uniformly charged ring of radius 1.2 m and total charge Q = -3 C placed on the origin of the x axis as shown in the picture below. a. Determine magnitude and direction of the electric...
-
Healthcare managers are involved in the annual budget and must have a good understanding of the financial statements and how they impact their individual departments. Capital budget proposals are...

Study smarter with the SolutionInn App


