Use a computer-aided, steady-state simulation program to estimate the K-values, using the P-R and S-R-K equations of
Question:
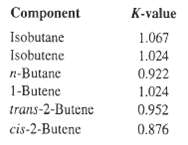
Use a computer-aided, steady-state simulation program to estimate the K-values, using the P-R and S-R-K equations of state, of an equimolar mixture of the two butane isomers and the four butane isomers at 220?F and 276.5 psia. Compare these values with the following experimental results [J. Chem. Eng. Data, 7, 331(1962)]:
Transcribed Image Text:
Component K-value Isobutane 1.067 Isobutene 1.024 n-Butane 0.922 1-Butene 1.024 trans-2-Butene 0.952 cis-2-Butene 0.876
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
Using the ChemCAD process simulation program the foll...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Roro Beach Shop is a shop in Pangkalan Balak, Melaka, that provides rental services for the following equipment: Equipment Buoy Canoe Speed Boat Water Scooter Equipment Code B S W Rental Rate/Hour...
-
An equimolar mixture of n-butane and n-hexane at pressure P is brought 10 a temperature of 95C, where it exists as a vapor/liquid mixture in equilibrium. If the mole fraction of n-hexane in the...
-
A steady stream of equimolar N2 and CO2 mixture at 100 kPa and 18C is to be separated into N2 and CO2 gases at 100 kPa and 18C. Determine the minimum work required per unit mass of mixture to...
-
Start with the partial model in the file Ch19 P08 Build a Model.xls on the textbooks Web site. Maggies Magazines (MM) has straight nonconvertible bond that currently yield 9%. MMs stock sells for $22...
-
Cost and fair value for the trading investments of Kootenay Ltd. at December 31, 2012, are $102,000 and $118,000, respectively. (a) Prepare the adjusting entry to record the investments at fair...
-
Carnival Corporation has recently placed into service some of the largest cruise ships in the world. One of these ships, the Carnival Breeze, can hold up to 3,600 passengers, and it can cost $750...
-
True or false: Zero to peak and peak readings are the same thing.
-
In Problem 2-3, if the direct manufacturing cost for cabinets is $27 and the rework cost is $8, compute the manufacturing cost per good product. Determine the manufacturing cost per product if the...
-
Calculate the ratio of the density at the centre of the Sun compared to the density of the photosphere. Density of Sun at the centre: 1.6 x 10 5 kg m -3 Density of the Sun in the photosphere: 2 x 10...
-
You have the following information for Brophy, Inc. December 31 Long-Term Debt ($ in millions) 7% debentures, $300 million face value, due Year 11, effective rate $14.6% Zero coupon bonds, $500...
-
Use the R-K equation of state to estimate the partial fugacity coefficients of propane and benzene in the vapor mixture of Example 2.5.
-
The disproportionation of toluene to benzene and xylenes is carried out in a catalytic reactor at 500 psia and 950?F. The reactor effluent is cooled in a series of heat exchangers for heat recovery...
-
In the context of Proposition 6.12, let \(t \in\{0,1, \ldots, T-1\}\) and \(A_{t} \in \mathscr{F}_{t}\) and suppose that, for some \(s \in\{t+1, \ldots, T\}\), there exists an event \(A_{s} \in...
-
During one of your instructor's lectures, take notes following the format described in the section "Compiling Fieldnotes." After the lecture, comment to yourself on selected notes in the left-hand...
-
Choose a function from a writing tool (e.g., columns or page layouts) and trace a brief history of it. Where did this technology or function come from? What does a person have to understand to know...
-
For each number \(x\) in the domain, the corresponding range value \(y\) is found by multiplying by 3 and then subtracting 5 .
-
Describe in detail what you actually do to plan for class projects (i.e., do you keep schedules and tasks "in your head," do you use a paper planner, do you create time lines or remind yourself with...
-
What does it mean to be socialized into the culture of an organization? How might we understand technical documents as socialization tools?
-
Let \((\mathcal{L},\langle\cdot, \cdotangle)\) be a Hilbert space and \(\mathcal{H}\) some linear subspace. Show that \(\mathcal{H}\) is a dense subset of \(\mathcal{L}\) if, and only if, the...
-
Give the products of the following reaction, where T is tritium: dioldehydrase Ad- CH CH3C-COH CoIII) coenzyme B12
-
Show that if p then the log odds-ratio is such that A-A'=(-p)/{(1-)}.
-
What is the difference between physical absorption and chemical (reactive) absorption?
-
In absorption, the absorbent is stripped to an extent that depends on its K-value. In stripping, the stripping agent is absorbed to an extent depending on its K-value. In Figure 6.1, it is seen that...
-
Can the degrees of freedom be determined for a hybrid system? If so, what is the easiest way to do it?
-
Reflect on a group interview you've had: * Was there anything you struggled with in preparing for the peer interviews? * What would you have changed about your interview? What would you have kept the...
-
Please conduct comprehensive research on the status of Metaverse, Choose a company, an industry, a product, or a service, and Share the strategies being used in Metaverse to develop a competitive...
-
Renovation and Restoration of community Park project For Risk management plan External risk : Budget Fluctuations in local economy Environment disasters Internal Risk: Safety Not enough money, staff...

Study smarter with the SolutionInn App


