Use Table 4.2 to predict whether the equilibrium for these reactions favors the reactants or the products.
Question:
Use Table 4.2 to predict whether the equilibrium for these reactions favors the reactants or the products.
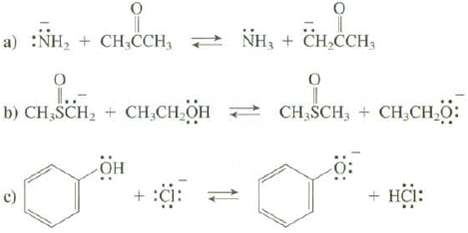
Transcribed Image Text:
a) :NH₂ + CH₂CCH, NH₂ + CH₂CCH, O 1..- b) CH₂SCH₂ + CH₂CH₂OH он + :C: 9 CH SCH, CH₂CH₂O: o + HCI:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Remember the equilibrium favors the formation of the weaker acid and the weaker base The ...View the full answer

Answered By

Shashikant Waghule
I worked as Online Tutor in Tecknit IT Enabled Services, Pune.
Currently working as Assistant Professor in Engineering College, but due to COVID-19 situation teaching online to Engineering students, helping students to solve their homework, assignments and mathematical problems.
Helping many students through social media to solve their maths problems.
Please give me chance to share my knowledge with SolutionInn students.
Thanking You.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Use Figure 16.3 to predict whether the equilibrium lies to the right or to the left in the following reactions:
-
Predict whether the following reactions will be spontaneous in acidic solution under standard conditions: (a) Oxidation of Sn to Sn2+ by I2 (to form I-) (b) Reduction of Ni2+ to Ni by I- (to form I2)...
-
Predict the products of the reactions of the following compounds with chromic acid and also with PCC. (a) Cyclohexanol (b) 1-methylcyclohexanol (c) Cyclopentylmethanol (d) Cyclohexanone (e)...
-
Why is it important to have a defined project scope? Why is it important to make sure there is agreement about the scope? Is there anything in the "Why Should You Use the WBS?
-
Mountain View Hospital has adopted a standard costing system for evaluation and control of nursing labour, Diagnosis Related Groups (DRGs) are used as the output measure in the standard costing...
-
Assume the following exchange rates: $2 = 1 in New York 410 = 1 in London 200 = $1 in Tokyo Indicate how profitable triangular, or three-point, arbitrage can take place.
-
A wing generates a lift \(\mathscr{L}\) when moving through sea-level air with a velocity \(U\). How fast must the wing move through the air at an altitude of \(10,000 \mathrm{~m}\) with the same...
-
1. Are there any losers with the TUI system described in this case? If so, who are they? Is this system justified despite creating losers? 2. Of the five competitive forces discussed in this case,...
-
Identify one Time Management tools that you use or would like to use. Explain your answer.
-
A Global private bank is aggressively looking to leverage technology to improve customer experience and reduce operational costs. Over the last few years, it has tied up with at least five startups...
-
Explain which compound is the weaker base. NH or NH NO b) or
-
Explain whether each of the following solvents would be acceptable for reactions involving this anion: (a) Liquid NH3 (b) CH3CH2OH (c)CH3CH2OCH2CH3 CHC=C: solvent
-
Suppose a limited number of tickets to a popular football game had to be rationed among the public. There are three ways of doing this: i. Auction: The tickets will go to the highest bidder. ii....
-
Jan Samson, CEO at CalleetaCO, sat staring at the now-empty boardroom. Her board of directors had reacted negatively to Jans growth proposals for expanding CalleetaCO globally, leaving Jan with a big...
-
Rudiger is sitting at his desk in his seventh-floor corner office in the city, gazing out over London and reflecting on life. At 43, he is at the top of his game. He has everything he could wish fora...
-
A dicing system has a first cost of $\$ 68,000$ with a life of 8 years and a salvage value of probably not more than $\$ 9,000$. The corporate MARR is $14 \%$. If the system has a maintenance cost of...
-
Meddevco (name changed) is a large multinational corporation that operates in the medical devices sector. The firm employs around 33,000 people in five divisions and has operations in 120 countries....
-
Compensation strategy includes how competitive the organization wants to be, the number of different compensation systems the organization wants to have, the mix of various reward and benefit...
-
In FRED, find and plot data on the ratio of outstanding total international debt to GDP for (a) the Euro area, (b) the United States, (c) Japan. These amounts represent the international indebtedness...
-
On October 1, 2021, Adoll Company acquired 2,600 shares of its $1 par value stock for $38 per share and held these shares in treasury. On March 1, 2023, Adoll resold all the treasury shares for $34...
-
Show that if a < b then a < (a + b)/2 < b.
-
When o-phthalaldehyde is treated with base, o-(hydroxymethyl) benzoic acid is formed. Show the mechanism of this reaction. CO2H 1. "OH 2. * " "CH2 o-Phthalaldehyde o-(Hydroxymethyl)benzoic acid
-
What is the stereochemistry of the pyruvate reduction shown in figure, does NADH lose its pro-R or pro-S hydrogen? Does addition occur to the Si face or Re face of pyruvate?
-
Assign R or S stereochemistry to the two chirality centers in isocitrate, and tell whether OH and H add to the Si face or the Re face of the double bond.
-
Assume that a firm has weighted average cost of capital of 3.6% with a corporate tax rate of 21%. The pre-tax cost of debt is 3.1%. In case the firm would have no debt, the cost of equity for the...
-
Consider the stock of Aya Company, which will pay an annual dividend of $3 one year from today. The dividend will grow at a constant rate of 3%, forever.The market requires a 12% return on the...
-
Analyze the video presented on the link below. Also, please discuss or explain the video and relate it to personal finance. https://www.youtube.com/watch?v=nFY0HBkUm8o

Study smarter with the SolutionInn App


