Use your knowledge of directing effects, along with the following data, to deduce the directions of the
Question:
Use your knowledge of directing effects, along with the following data, to deduce the directions of the dipole moments in aniline andBromobenzene.
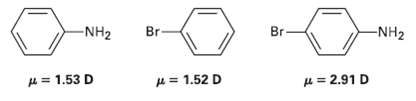
Transcribed Image Text:
Br- -NH2 Br- -NH2 H = 1.52 D H = 2.91 D A= 1.53 D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Br 153 D Br has a strong electronwithdrawing induct...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Use your knowledge of non-Western cultures to draw attention to differences in non-verbal communication between one or more of these cultures and Western (as represented, for example, by Australia)...
-
Use the Mann-Whitney U test and the following data to determine whether there is a significant difference between the values of group 1 and group 2. Let = .05. Group 1 Group 2 15 ........ 23 17...
-
Use the Mann-Whitney U test and the following data to determine whether there is a significant difference between the values of group 1 and group 2.Let = .05. Group 1 .. Group 2 15 ..... 23 17...
-
Which statements when inserted independently will throw an exception at runtime? (Choose two.) var x = new LinkedList (); x.offer (18); // INSERT CODE HERE A. B. x. peek (); x.peek(); x. poll ();...
-
Sam and Abby are dependents of their parents, and each has income of $2,100 for the year. Sam's standard deduction for the year is $1,050, while Abby's is $2,450. As their income is the same, what...
-
With reference to Exercise 2.3, calculate the quartiles. In Exercise 2.3 A student at the University of Wisconsin surveyed 40 students in her dorm concerning their participation in extracurricular...
-
Determine whether the random variable x is discrete or continuous. Explain your reasoning. (a) Let x represent the number of lightning strikes that occur in Wyoming during the month of June. (b) Let...
-
An investor purchased the following 5 bonds. Each bond had a par value of $1,000 and an 8% yield to maturity on the purchase day. Immediately after the investor purchased them, interest rates fell...
-
Question 1. From the research for this terms Crisis Management Team Assignment - Taylor swift and Ticket Master Crisis a. Explaining, with some detail, three (3) specific things that the leader(s) in...
-
Explain the stages of the consumer buyer decision process and describe how you or your family went through this process to make a recent purchase.
-
Benzyl bromide is converted into benzaldehyde by heating in dimethyl sulfoxide. Propose a structure for the intermediate, and show the mechanisms of the two steps in thereaction. CH2BR E2 reaction...
-
Identify the reagents represented by the letters a?c in the following scheme: Br Br Br Br
-
Milton Company has just received the following monthly bank statement for June 2012. Data from the cash account of Milton Company for June are as follows: At the end of May, Milton had three checks...
-
Directions: Read the newsletter Make Your Own Job. Complete the activities in the newsletter. These activities will not be submitted for grading. http://www.ogburn.org/PFLesson15.pdf Explain the...
-
1. Calculate the equilibrium constant, Keq, for the following reaction at 25 C, if [NO]eq = 0.106 M, [02]eq = 0.122 M and [NO2]eq = 0.129 M. 2 NO (g) + O2 (g) 2 NO2 (g) 2. Find the equilibrium...
-
LO 1 LO 1 11. Valuing Preferred Stock E-Eyes.com has a new issue of preferred stock it calls 20/20 preferred. The stock will pay a $20 dividend per year, but the first dividend will not be paid until...
-
4. Solve the following systems: (a) x + x2 + 2x3 = 8 - -x1 2x + 3x3 1 3x17x2 + 4x3 = 10 (b) x+y+z=3 4x + 4y + 4z = 7 3xy + 2z = 5
-
Shown below is the block diagram of the datapath that was used for Greatest Common Divisor (GCD) calculation in the lab task. 19 else selA SelB LoadA LoadB AeqB else AltB Loadgcd done Y = A-B; end if...
-
Recommend additional information that Zik could provide to enhance his portfolio reports for Patel. Sharfepto Zik, a private wealth manager, is meeting with a client, Garbanzo Patel, in order to...
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
a. An automobile air conditioner uses the vapor compression refrigeration cycle with HFC-134a as the refrigerant. The operational temperature of the evaporator is 7 C and that of the condenser is 45...
-
Suggest explanations for the origins of "ibu," "pro," and "fen" in the name ibuprofen. Provide a systematic name for thiscompound OH O,N. NO2 NO2 Picric acid
-
The pK a for the picric acid is 0.42. Explain why it is such a strong acid.
-
To find a base that is strong enough to deprotonate benzoic acid but not p-methyl phenol. Then explain how this base might be used to separate these two compounds in the laboratory.
-
4. The water in Earth's atmosphere blocks most of the infrared waves coming from space. In order to observe light of this wavelength, the Kuiper Airborne Observatory has been developed. The...
-
What amount of gain should Pharoah recognize on the exchange assuming lack of commercial substance?
-
If R1=7 Q, R2=3 Q, R3=9 Q, V= 12 V, and V2=8 V. Find the current (in A) that pass through R. R R R V V 2 +

Study smarter with the SolutionInn App


