Using Equation 18.36 and the results of Problem 18.33, determine the temperature at which the electrical conductivity
Question:
Using Equation 18.36 and the results of Problem 18.33, determine the temperature at which the electrical conductivity of intrinsic germanium is 22.8 (?-m)???1.
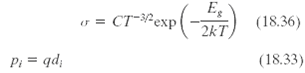
Transcribed Image Text:
Eg v = CT=&exp(-) ст-ехр (18.36) 2kT (18.33) Pi = qd;
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
This problem asks that we determine the temperature at which the el...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Determine the temperature at which 5 percent of diatomic oxygen (O2) dissociates into monatomic oxygen (O) at a pressure of 3 atm.
-
Determine the temperature at which 2% of diatomic oxygen (O2) dissociates into monatomic oxygen (O) at a pressure of 3 atm.
-
The results of Problem 61 apply also to an electrostatic precipitator (Figures 25.30 and P25.61). An applied voltage V = Va - Vb = 50.0 kV is to produce an electric field of magnitude 5.50 MV/m at...
-
Please watch the video link and answer the following questions. https://youtu.be/VtvjbmoDx-I On January 22,1984, Apple produced one of the most famous Super Bowl ads of all time. Directed by Ridley...
-
Find the following values assuming a regular, or ordinary, annuity: a) The present value of $400 per year for ten years at 10 percent b) The future value of $400 per year for ten year at 10 percent...
-
Exercise 9 described the formation of glucose in plants, which take in CO2 and H2O and give off O2. Is this process exothermic or endothermic? If exothermic, where does the energy go? If endothermic,...
-
The price of a bond issued by C & M plc is 85.50 per cent of par value. The bond will pay an annual 8.5 per cent coupon until maturity (the next coupon will be paid in one year). The bond matures in...
-
Review the data from Great Fender given in E23-19. Consider the following additional information: Great Fender allocates manufacturing overhead to production based on standard direct labor hours....
-
10:14 AM You sent 17010 Vina Santiago, the owner of ALOHA RESTO, is engaged in restaurant business and classified as non-VAT taxpayer with TIN no. 123-456-789 registered with RDO 98 (Cagayan de Oro)....
-
Kitchen Works Ltd is a carpentry business that specializes in the production of kitchen cabinets. The company budgeted to produce 44,000 kitchen chairs for the month of January, but actually produced...
-
At temperatures near room temperature, the temperature dependence of the conductivity for intrinsic germanium is found to equal where C is a temperature-independent constant and T is in Kelvins....
-
Estimate the temperature at which GaAs has an electrical conductivity of 3.7 3 1023 (V-m)21 assuming the temperature dependence for of Equation 18.36. The data shown in Table 18.3 might prove...
-
A pump with an 8-in diameter impeller is operating at 1750 rpm and delivering 40 gpm with a total dynamic head of 55 ft. The pump's brake horsepower (BHP) is 1.2 hp under these conditions. Assume...
-
The following information about the fixed assets for Martinez Books Inc. has been provided to you. Martinez uses straight-line depreciation. The assets were acquired on January 1 and have no residual...
-
d Wiater Company operates a small manufacturing facility. On January 1, 2021, an asset account for the company showed the following balances: Equipment Accumulated Depreciation (beginning of the...
-
A company operates a job costing system. The company s standard net profit margin is 2 0 % of sales value. The estimated costs for job B 1 2 4 are as follows: Direct material 3 kg @ $ 5 per kg Direct...
-
Palisade Creek Co. is a merchandising business that uses the perpetual inventory system. The account balances for Palisade Creek Co. as of May 1, 2016 (unless otherwise indicated), are as follows:...
-
Y ou push a 1 5 . 0 kg lawnmower across the lawn at constant speed. To do so , you exert 1 2 0 N of force along the handle, which makes an angle of 3 5 degree with the horizontal; ( a ) Draw the...
-
Under PKPA, when must a state enforce the decree of another state?
-
Graph one period of each function. y = 4 cos x
-
Sketch the hypocycloid x 2/3 + y 2/3 = 1 and find the volume of the solid obtained by revolving it about the x-axis.
-
Why might it be desirable to weaken the bond between a reinforcing fiber and a ceramic-matrix material?
-
Discuss some of the techniques used to cut fiber-reinforced composites.
-
What is the major concern when considering the joining of fiber-reinforced composites?
-
5. A friend has been offered a sales repre c., a small publisher of computer-related books, but wants to know more about the com- pany. Because of your expertise in financial analysis, you offer to...
-
When employees try to determine whether their current pay is equitable or inequitable, they generally compare their pay: to employees who appear to be the boss's favorites. to objective industry -...
-
1. You are an operation manager in shoe manufacturing, and your boss requests you to have the analysis for expanding. Please discuss the role of logistics variables in the decision as to where to...

Study smarter with the SolutionInn App


