Using the curved-arrow notation, derive a resonance structure for the allyl anion (shown here) which shows that
Question:
Using the curved-arrow notation, derive a resonance structure for the allyl anion (shown
here) which shows that the two carbon-carbon bonds an identical bond order of 1.5 and that the unshared electron pair (and negative charge) is shared equally by the two terminal carbons.
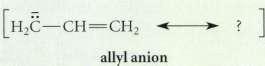
Transcribed Image Text:
H2C CH CH2 allyl anion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (15 reviews)
H CHCH ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In each of the following sets, show by the curved-arrow or fishhook notation how each resonance structure is derived from the other one, and indicate which structure is more important and why. |.-N ]...
-
Use the curved-arrow or fishhook notation to derive the major resonance structures for each of the following species. Determine which, if any, structure is the most important one in each case. (a)...
-
The resonance structures of carbon monoxide are shown below. Show how each structure can be converted into the other using the curved-arrow notation. :C-: :C=0:
-
A department store is being planned for a new shopping mall. Using the information in Table 4.8, assign departments to locations in order to minimize traffic flow through the store. TABLE 4.8...
-
How does climate influence mass wasting? Also, intuitively, it would seem that rainy climates would be most susceptible to mass wasting. But why might arid climates experience a great deal of mass...
-
Bangura, Inc. has regular taxable income of $932,500,000 and AFSI of $1,437,100,000. Compute Banguras regular tax liability, tentative minimum tax, AMT (if any) and total tax due.
-
Two other important symbols of Rastafarian culture are ganja (marijuana) and the use of a special vocabulary. The use of ganja has been common on Jamaican agricultural estates since the turn of the...
-
Pisa Pizza Parlor is investigating the purchase of a new $45,000 delivery truck that would contain specially designed warming racks. The new truck would have a six-year useful life. It would save...
-
Assume you are a staff associate with GKN Associates assigned to the Mystery, Inc. engagement. John Cole, the senior associate on the job, has asked for your assistance in performing journal entry...
-
The Protek Company is a large manufacturer and distributor of electronic components. Because of some successful new products marketed to manufacturers of personal computers, the firm has recently...
-
Arrange the compounds in each of the following sets in order of decreasing pKa, highest first. Explain your reasoning. (a) CLCH2CH2SH CH3CH2OH CH3CH2SH (b) CH,CH,OH (CH3),N-CH-CH,OH (CH3)N OH
-
Using Table 3.1, as well as the data given below, estimate the equilibrium constants for the following reactions at 25C. pK, 10.5
-
Find the indicated quantity for an infinite geometric series. S = 4 + 2/2, r = ILS || ?
-
A cargo plane is moving with a horizontal velocity of vx = + 2 3 1 m / s at a height of y = 1 1 7 0 m above level ground when it releases a package. Ignoring air resistance, how much time, in...
-
12. Compute the time needed to bake the cake (use Newton's Law of Cooling) if the initial temp of the mix before baking was 62 degrees and the oven temperature is set at 350 degrees. Assume that 10...
-
After evaporating metal on a semiconductor with an effective mass of 0.1 mo, you apply a forward bias of V=0.2 V. You measure a current of 492.5 A at T=300 K and 73.5 A at T=250 K. What is the...
-
15. Nichrome wire of cross-sectional radius 0.791 mm is to be used in winding a heating coil. If the coil must carry a current of 9.25 A when a voltage of 1.20 x 10 V is applied across its ends, find...
-
A gardener uses a shovel as a lever to lift a 200 N rock a distance of 0.20 meters. He does this by applying 50 N of force to the end of the shovel. a) Calculate the mechanical advantage of the...
-
Discuss health care concerns in retirement.
-
How does Kant answer Humes bundle theory of self? Do you think he is successful?
-
Determine whether these structures represent the same compound or isomers: ext0Q L a) O b) d) CH3 e) CH3CHCHCHCH3 CHCHCHCHCH 3 CH, f) CHCHCHCHCHCH3 CHCHCHCHCHCH, CH3 CH3 ~
-
Calculate the DU for these formulas and draw two constitutional isomer for each: (a) C 10 H 20 O (b) C 6 H 9 N (c) C 7 H 14 F 2 (d) C 6 H 8 CIN (e) C 9 H 15 NO
-
Determine the DU for each of these structures: Jesen OCHCH3 c) Br COH -Og OCCH3 Acetylsalicylic acid (aspirin) b) N NH, O CH-CH-C-OH H Tryptophan (an amino acid)
-
Joey set up a lawn-mowing business in his neighborhood. He currently has 7 customers that want their lawns mowed each week, for which he charges $25.00 each. Joey spends $10.00 a week in gas and...
-
Vanessa has an outstanding bond with a 7.10 % semi-annual coupon and has 16 years left to mature. The current market rate is 10.50% per annum. What is the current yield of Vanessa's Bond.
-
Estimate the hydrogen consumption required to completely remove the sulfur from a hydrotreater feedstock and to reduce the nitrogen content of the product to 15 ppm by weight. The 48.5 API naphtha...

Study smarter with the SolutionInn App


