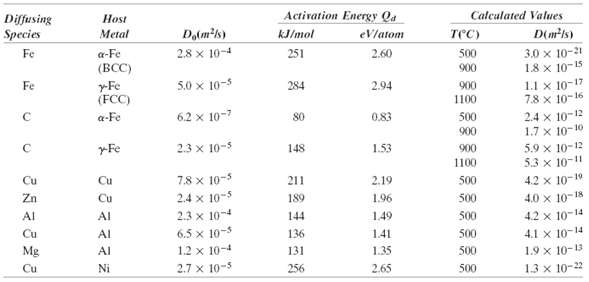
Using the data in Table 5.2, compute the value of D for the diffusion of zinc in
Question:
Using the data in Table 5.2, compute the value of D for the diffusion of zinc in copper at 650?C.
Transcribed Image Text:
Activation Energy Qa Calculated Values Diffusing Species Host Do(mk) Đ(mts) Metal kJ/mol тес) eVlatom 3.0 x 10-21 1.8 x 10-15 1.1 x 10-17 7.8 x 10-16 a-Fe (BCC) Fe 2.8 x 10-4 251 2.60 500 900 5.0 x 10-5 Fe y-Fe (FCC) 284 2.94 900 1100 2.4 x 10-12 1.7 x 10-10 6.2 x 10-7 a-Fe 80 0.83 500 900 5.9 x 10-12 5.3 x 10-11 yFe 2.3 x 10-5 1.53 148 900 1100 7.8 x 10-5 Cu 4.2 x 10-19 Cu 211 2.19 500 Zn 2.4 x 10-5 500 4.0 x 10-18 Cu 189 1.96 2.3 x 104 1.49 500 4.2 x 10-14 Al AI 144 6.5 x 10-5 1.2 x 10-4 4.1 x 10-14 Cu AI 136 141 500 1.9 x 10-13 1.3 x 10-22 Mg Al 131 1.35 500 2.7 x 10-5 Cu Ni 256 2.65 500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
Incorporating the appropriate data from Table 52 into Equation 58 leads to 1 In the lefthand window ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Using the data in Table 13.2, ask whether peoples first guess is usually better than their second guess. (This parallels advice that you often receive about test taking to the effect that you should...
-
Using the data in Table 12.29, perform a multiple comparisons procedure to identify which specific underlying means are different? Nutrition Researchers compared protein intake among three groups of...
-
Using the data in table 6-11, calculate a three-month moving average forecast for month 12.
-
EFG Manufacturing, a private company, produces light bulbs. It has 100 employees. 15 of their employees have asked for short breaks during the workday for religious purposes. They have also asked for...
-
Julius W. Erving (Dr. J) entered into a four year contract to play exclusively for the Virginia Squires of the American Basketball Association. After one year, he left the Squires to play for the...
-
Refer to all of the facts in Problem 12-6A. Required 1. Prepare a statement of cash flows for 2012 using the indirect method in the Operating Activities section. 2. On the basis of your statement in...
-
1. Recent consumer surveys suggest that the three most important qualities of a retail operation are price, product (quality and selection), and convenience. Based on that information, describe how...
-
Knight, Corp., a lamp manufacturer, provided the following information for the year ended December 31, 2012: Requirements 1. Prepare a schedule of cost of goods manufactured. 2. What is the unit...
-
Explain the concept of paging in virtual memory. How do page tables work, and what are the differences between single-level and multi-level page tables in terms of memory usage and translation...
-
On January 2, Year 1, Poplar Ltd. purchased 80% of the outstanding shares of Spruce Ltd. for $2,000,000. At that date, Spruce had common shares of $500,000 and retained earnings of $1,250,000 and...
-
Cite the values of the diffusion coefficients for the inter-diffusion of carbon in both -iron (BCC) and -iron (FCC) at 900C. Which is larger? Explain why this is the case.
-
At what temperature will the diffusion coefficient for the diffusion of copper in nickel have a value of 6.5 ? 10 -17 m 2 /s. Use the diffusion data in Table 5.2. Diffusing Species Host Activation...
-
Statement of Legal Debt Margin. In preparation for a proposed bond sale, the city manager of the City of Appleton requested that you prepare a statement of legal debt margin for the city as of...
-
You have been given a set of physical DFDs for a new system to review for acceptability. What questions should you ask yourself when reviewing them?
-
How are outputs classified?
-
What is a well-designed source document?
-
What are some of the characteristics of prototypes?
-
If there is no forwarding, what new inputs and output signals do we need for the hazard detection unit in Figure 4.60? Using this instruction sequence as an example, explain why each signal is...
-
We introduced Tennessee's Project STAR (Student/Teacher Achievement Ratio) in Exercise 2.22. The data file is star5_small. [The data file star5 contains more observations and more variables.] Three...
-
Government is advised to tax goods whose demand curves are inelastic if the goal is to raise tax revenues. If the goal is to discourage consumption, then it ought to tax goods whose demand curves are...
-
A certain ammeter has a resistance of 5.00 x 10 -5 on its 3.00-A scale and contains a 10.0- galvanometer. What is the sensitivity of the galvanometer?
-
Give the approximate minimum temperature at which it is possible to austenitize each of the following iron-carbon alloys during a normalizing heat treatment: (a) 0.20 wt% C, (b) 0.76 wt% C, and (c)...
-
Give the approximate temperature at which it is desirable to heat each of the following iron-carbon alloys during a full anneal heat treatment: (a) 0.25 wt% C, (b) 0.45 wt% C, (c) 0.85 wt% C, and (d)...
-
What is the purpose of a spheroidizing heat treatment? On what classes of alloys is it normally used?
-
Explain how probability can be applied to decision-making in public administration situations.
-
Do you think that all levels of management have the same functional skills needed for the workplace? How do managers in organizations communicate in terms of tone and leadership style?
-
Accustart Ltd. acquired 38% of the common shares of Lecce Ltd. on January 1, 2024, by paying $5.76 million for 144,000 shares. Lecce declared a cash dividend of $0.60 per share in each quarter that...

Study smarter with the SolutionInn App


