Using the information available in Figure 4.2, predict the position of the equilibrium in these reactions; that
Question:
Using the information available in Figure 4.2, predict the position of the equilibrium in these reactions; that is, predict whether there is a higher concentration of reactants or products present at equilibrium:
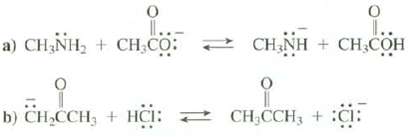
Transcribed Image Text:
0 a) CHÍNH, + CH CO: 1↓ O CH₂NH + CH₂COH 0 b) CH₂CCH₂ + HCI: CH₂CCH₂ + C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
The equilibrium favors the formation of the more stable compounds In the ...View the full answer

Answered By

Poonam Chaudhary
I have 15 month+ Teaching Experience
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A statistics teacher is wondering whether there is a relationship between the marks of students in their first-year business math course, and their marks in the second-year statistics course....
-
Using the information available at the SEC's website or any other authoritative source, describe how the sec is structured.
-
Based on the information available in the case, discuss what you think are the reasons for people's resistance to the MRP implementation. Discuss if there is any value to the task force idea that was...
-
Why is Supplier Relationship is important to a business. explain
-
The 'Real life' on page 504 explains how standard costing systems may be adopted to better suit managers' decision needs. This includes using different definitions of standard costs to suit different...
-
Let X~N (10, 25). Compute (a) P (X 20) (b) P (X > 5) (c) P (12 X 15) (d) P (|X12| 15).
-
Describe the evolution of the Developmental Configuration and its baseline.
-
A successful businessman in the community has contacted the Moose County Board of Commissioners about donating income producing securities to the County to support a particular activity. Under the...
-
Make observations and analyze them - Begin the observation session(s) well in advance of the due date. You will need to make at least two 30- minute observations of your subject. The first...
-
A store maintains data on customers, products and purchase records in three tables: CUSTOMER, PRODUCT, PURCHASE. The store manager wants to know which product is on its maximum discount for each...
-
Indicate whether these species are weaker or stronger bases than hydroxide ion. The Ka is or pKa values are for the conjugate acids. a) :NH, (K = 10-38) c) NH, (PK, = 9.24) b) CHCHCH d) :CI: (pK =...
-
Use the information in Figure 4.2 to predict the positions of the equilibria in the reactions in problem 4.4.
-
Rank in order, from largest to smallest, the currents I a to I d through the four resistors in FIGURE Q28.1. + 2V - + 1V - 2 + 2V - 1 + 1V - 1 Ia FIGURE Q28.1
-
Q3:(10 Pnts) Given Caesium chloride (CsCl) crystal with lattice constant 4.11A. CsCl takes Body center cubic in the [100] direction. The material build in the [111] direction and looks like the given...
-
Job 1 233 Processing Time Due Date in Days 7 152 (day) 8 12 6 4 18 5 6 17) Using the data in the table above a) (6 points) Sequence jobs according to SPT. b) (6 points) Find the average cycle time...
-
II. What Do the Experts Say? 4 3. According to the automotive engineer, what two quantities will you need to compare in order to decide whether the driver could have prevented the accident? According...
-
Interference fringes are produced by monochromatic light of wavelength 5 4 6 0 A . When a thin transparent sheet of thickness 6 . 3 x 1 0 cm is introduced in the path of one of the interfering beams,...
-
A spring has an unstretched length of 1 0 . 0 cm . A 2 5 g mass is hung from the spring, stretching it to a length of 1 5 . 0 cm . Once you determine the spring constant, you replace the mass with...
-
Catherine Rampell is an economics writer for the Washington Post. In an article in 2023, she observes that the average U.S. tariff rate on mens underwear is 11.5 percent. The average rate on womens...
-
To balance the chemical equation SiH3 + O2 SiO2 + HO, you could introduce coefficients a, b, c, d and write aSiH3 + bO2 cSiO + dHO then write linear equations for each element. The equation for Si...
-
Graph the function y = x 3 150x.
-
Draw an energy diagram for a two-step reaction with Keq > 1. Label the overall G, transition states, and intermediate. Is G positive or negative?
-
Draw an energy diagram for a two-step exergonic reaction whose second step is faster than its first step.
-
Draw an energy diagram for a reaction with Keq = 1. What is the value of G in this reaction?
-
An examination of the cash activities during the year shows the following. GREAT ADVENTURES Cash Account Records July 1 , 2 0 2 4 , to December 3 1 , 2 0 2 4 Cash Receipts Cash Disbursements Date...
-
Given the data below answer the question. E F G H J Inventory Turnover Ratio Ending Inventory (Dec. 31st) $50,000 Month Ending Inventory Cost of Goods Sold Inventory Turnover Ratio Formulas January...
-
[3] 4. In 1995, Mary bought an antique chair for $55. It is estimated that the chair will increase in value by 1.8%/year. a) Find the current value of the chair. 121 [2] b) In what year will the...

Study smarter with the SolutionInn App


