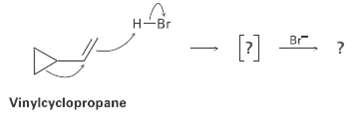
Vinylcyclopropane reacts with HBr to yield a rearranged alkyl bromide. Follow the flow of electrons as represented
Question:
Vinylcyclopropane reacts with HBr to yield a rearranged alkyl bromide. Follow the flow of electrons as represented by the curved arrows, show the structure of the carbocation intermediate in brackets, and show the structure of the final product.
Transcribed Image Text:
H-Br Br [] Vinylcyclopropane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
Br CH3 Y Y HBr CH3 Attack of the electrons of ...View the full answer

Answered By

Hassan Ali
I am an electrical engineer with Master in Management (Engineering). I have been teaching for more than 10years and still helping a a lot of students online and in person. In addition to that, I not only have theoretical experience but also have practical experience by working on different managerial positions in different companies. Now I am running my own company successfully which I launched in 2019. I can provide complete guidance in the following fields. System engineering management, research and lab reports, power transmission, utilisation and distribution, generators and motors, organizational behaviour, essay writing, general management, digital system design, control system, business and leadership.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A Follow the flow of electrons indicated by the curved arrows in each of the following reactions, and predicts the products thatresult: (b) :0- (a) -: "CH
-
Use the curved-arrow notation to indicate the flow of electrons in each of the transformations given in Fig. P3.33. (a) (b) (c) (d) (CHJ.NH-CH.-CH-C-OC-H5 BrC2 HC CH CH3 CH2 CH CH C: T:0:
-
Normally, the flow of electrons to O 2 is tightly linked to the production of ATP via the electrochemical gradient. If ATP synthase is inhibited, for example, electrons do not flow down the...
-
(a) Write an equation describing a sinusoidal transverse wave traveling on a cord in the positive direction of a y axis with an angular wave number of 60 cm-1, a period of 0.20 s, and an amplitude of...
-
1. To what extent has Julia chosen the right approach to leading the managers in her unit of the financial services firm? 2. What advice can you offer Julia to be a more effective leader? 3. What...
-
The following are some of the most important trigonometric identities. Check them at a. = 0, b. = /4, c. = /2, d. = . sin2() + cos2() = l.
-
How are project supply chains different from the make-to- stock supply chains of the traditional manufacturing organizations?
-
The comparative balance sheet of House Construction Co. for June 30, 2010 and 2009, is as follows: The income statement for the year ended June 30, 2010, is as follows: The following additional...
-
1) Who are the two sides involved in the case? Who is the defendant, and who is the plaintiff? 2) What is the issue that is being dealt with in the case? What Constitutional question does it deal...
-
1). Compute return on equity (ROE), return on assets (ROA), and return on financial leverage (ROFL) for the most recent year. Assume a marginal statutory tax rate of 30%. 2) Disaggregate the ROAs...
-
Addition of HCI to 1-isopropcnyl-1-methylcyclopcntane yields 1-chloro-1, 2, 2- trimethylcyclohexane. Propose a mechanism, showing the structures of the intermediates and using curved arrows to...
-
Calculate the degree of unsaturation in each of the following formulas: (a) Cholesterol, C27H46O (b) DDT, C14H9C15 (c) Prostaglandin E1, C20H34O5 (d) Caffeine, C8H10N4O2 (e) Cortisone, C21H28O5 (f)...
-
On January 23, 2006, CNET News.com quoted an anonymous source describing strategic plans made at a meeting of HPs board of directors. Because the meeting was held behind closed doors and with a...
-
Consider the following implementation of an iterative method: from numpy import cos, sin, log. x = 0.5 err = abs (cos(x)+log(x)) while err > 1e-6: dx = (cos(x)+log(x))/(-sin(x) +1/x) x += - dx err =...
-
Tr Exercise 2.24. Consider a two-state Markov chain with state space {0, 1} and the following transition probability matrix: 1 - P = q 1-9 For all choices of p, q, [0, 1] find all stationary...
-
The carrying case would be sold to the end consumer for $ 4 9 . 9 9 . Lisa will not sell directly to the consumer but will use a wholesaler who will sell to a retailer ( e . g . , university...
-
Shandra has collected data about how the price of a used Volkswagon Golf decreases as the Volkswagon Golf ages. Shandra would like to use a trendline to predict the price of a used Volkswagon Golf...
-
Hazelnut Corporation manufactures lawn ornaments. It currently has two product lines, the basic and the luxury. Hazelnut has a total of $169,122 in overhead. The company has identified the following...
-
The highest dew point ever recorded was 95F, recorded in Saudi Arabia. Was the air humid or dry at that time? Explain your reasoning.
-
Test whether the 5-year survival rate for breast cancer is significantly different between African American and Caucasian women who are younger than 50 years of age and have localized disease....
-
Examine the heating curve for water in Section 12.7 (Figure 12.36). Explain why the curve has two segments in which heat is added to the water but the temperature does not rise. Temperature (C) 125...
-
Draw a Newman projection of the confirmation of (2S,3R,4S)-2,3,4-hexanetriol (shown above) About the C2{3 bond, with C2 nearest the observer
-
Draw (a) Sawhorse projections and (b) Line-and-wedge structures for all three staggered conformations of butane.
-
Draw a sawhorse projection and a line-and-wedge structure for the conformation of (2S,3S)-2,3-dibromobutane in which the methyl groups are anti.
-
There are n undergraduate students and k departments at some university. The Student Senate must have k students, one from each department. It also should have k freshmen, k sophomores, kg juniors,...
-
what are Total Liabilities equate to 478,523,226? Debt to Capitalization Ratio = (Total Liabilities (Total Assets + Total Equity)) x 100 Debt to Capitalization Ratio =...
-
Victor Mineli, the new controller of Wildhorse Co., has reviewed the expected useful lives and salvage values of selected depreciable assets at the beginning of 2025. Here are his findings: Type of...

Study smarter with the SolutionInn App


