We?ll look at cycloalkanes?saturated cyclic hydrocarbons? and we?ll see that the molecules generally adopt puckered, non-planar conformations.
Question:
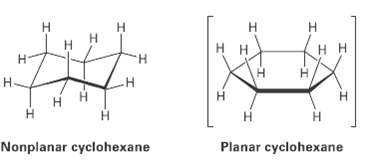
We?ll look at cycloalkanes?saturated cyclic hydrocarbons? and we?ll see that the molecules generally adopt puckered, non-planar conformations. Cyclohexanc, for instance, has a puckered shape like a lounge chair rather than a flat shape. Why?
Transcribed Image Text:
Н н H. H. H. H H Н H. Н- Н H. Н Planar cyclohexane Nonplanar cyclohexane I-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
A puckered ring allows all the ...View the full answer

Answered By

Shubham gupta
This is Swetha. I have completed Bachelors degree in 2016 and my background is Electronics and communication Engineering.
I have four years experience in tutoring.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Why do we focus on cash flows rather than accounting profits in making our capital- budgeting decisions? Why are we interested only in incremental cash flows rather than total cash flows?
-
Why do economists generally favor vouchers rather than public production or subsidies to achieve an efficient outcome?
-
Why is automation generally regarded as evolutionary rather than revolutionary? Explain.
-
Consider examples 9.2 and 9.3. Assume that social damage is quadratic as in the examples. Following section 9.2.3, assume that the regulator can only set a uniform tax. Determine the formula for the...
-
In the prospectus for the Brazos Small Cap Fund, the fee table indicates that the fund has a 12b -1 fee of 0.35 percent and an expense ratio of 1.65 percent that is collected once a year on December...
-
Where do the components of the changes in retained earnings appear in the statement of cash flows? Assume the indirect method is used to prepare the statement of cash flows.
-
The Azuza Company is liquidating. After paying off all of its creditors, the company has \(\$ 1.5\) million to distribute between its preferred stockholders and its common stockholders. The aggregate...
-
Sawyer Company was formed on December 1, 2013. The following information is available from Sawyers inventory records for Product BAP. A physical inventory on March 31, 2014, shows 3,200 units on...
-
4 Part 2 of 5 10 points eBook Print O References Required information [The following information applies to the questions displayed below.] The following transactions apply to Walnut Enterprises for...
-
A variable dielectric capacitive displacement sensor consists of two square metal plates of side 5 cm, separated by a gap of 1 mm. A sheet of dielectric material 1 mm thick and of the same area as...
-
The cholesterol-lowering agents called statins, such as simvastatin (Zocor) and pravastatin (Pravachol), are among the most widely prescribed drugs in the world. Identify the functional groups in...
-
We?ll see that there are two isomeric substances both named 1, 2-dimethylcyclohexane. Explain. -C3 1,2-Dimethylcyclohexane CH
-
Under what circumstances might inventory turnover be too high? That is, what possible negative consequences might occur?
-
What is asymmetric information and what is its significance in the relationship between a bank and a borrower?
-
What conditions must exist before a grandfather, father, son backup strategy can be used? Briefly discuss the advantages and disadvantages of the strategy.
-
Explain cash management.
-
Briefly describe the various types of logs that can be used for recovery purposes. Why might a combination of logging strategies be used for recovery purposes?
-
Briefly describe the three major types of exposure in the communication subsystem.
-
The design team for Superior Electronics is creating a mobile audio player and must choose between two design alternatives. Which is the better environmental design alternative, based on achieving a...
-
Eleni Cabinet Company sold 2,200 cabinets during 2011 at $160 per cabinet. Its beginning inventory on January 1 was 130 cabinets at $56. Purchases made during the year were as follows: February . 225...
-
With reference to Figure 12-20, which is the more volatile liquid, benzene or toluene? At approximately what temperature does the less volatile liquid have the same vapor pressure as the more...
-
Starting with the compound or compounds indicated in each part and using any other needed reagents, outline syntheses of each of the following compounds. (You need not repeat steps carried out in...
-
How would the molecular ion peaks in the respective mass spectra of CH3Cl, CH2 Cl2, CHCl3, and CCl4 differ on the basis of the number of chlorines? (Remember that chlorine has isotopes 35Cl and 37Cl...
-
Provide the reagents necessary for the following synthetic transformations. More than one step may be required. (a) (b) (c) (d) (e) (f) OCH3 Br
-
The Financial controller of "BESTH Co." is about to select among three available projects. He was provided the following information: Project Rate of return X 12.10 % Y 11.80% Z 12.95% BESTH Co. has...
-
Dickson Corporation is comparing two different capital structures. Plan I would result in 35,000 shares of stock and $100,500 in debt. Plan II would result in 29,000 shares of stock and $301,500 in...
-
The commute time for people in a city has an exponential distribution with an average of 0.5 hours. What is the probability that a randomly selected person in this city will have a commute time...

Study smarter with the SolutionInn App


