What alkyl halides might the following alkenes have been madefrom? (b) C CH (a) (a) CH CHH2H2%3DCH2
Question:
What alkyl halides might the following alkenes have been madefrom?
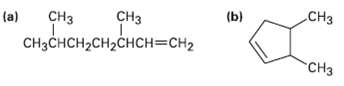
Transcribed Image Text:
(b) CНз CHз (a) (a) CHз CHзснсH2сH2снсн%3DCH2 "CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Strategy For maximum yield the alkyl halide reactan...View the full answer

Answered By

Sandip Nandnawar
I am a B.E (Information technology) from GECA and also have an M.C.M from The University of RTMNU, MH.
I worked as a software developer (Programmer and TL). Also working as an expert for the last 6 years and deal with complex assessment and projects. I have a team and lead a team of experts and conducted primary and secondary research. I am a senior software engg and senior expert and deal with all types of CSE and IT and other IT-related assessments and projects and homework.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Alkyl halides add to alkenes in the presence of AlCl3; yields are the highest when tertiary halides are used. Predict the outcome of the reaction of tert-pentyl chloride (1-chloro-2,...
-
What alkyl halides would you utilize to synthesize the following compounds, using the organoborane shown? a. b. c.
-
Alkyl halides have been used as insecticides since the discovery of DDT in 1939. DDT was the first compound to be found that had a high toxicity to insects and a relatively low toxicity to mammals....
-
A small project consists of three activities: A, B, and C. To start activity C, both activities A and B need to be complete. Activity A takes 3 days with a probability of 50 percent and 5 days with a...
-
What does the opening vignette tell you about what makes an employee stay in a job, despite difficulties?
-
Figure 5.3 illustrates the Bayesian belief network for the data set shown in Table 5.3. (Assume that all the attributes are binary). (a) Draw the probability table for each node in the network. (b)...
-
Use a process simulator to model a two-stage compression system with an intercooler. The feed stream consists of \(95 \mathrm{~mol} \%\) hydrogen and \(5 \mathrm{~mol} \%\) methane at \(100^{\circ}...
-
Opportunity costs. The Wolverine Corporation is working at full production capacity producing 10,000 units of a unique product Rosebo. Manufacturing cost per unit for Rosebo is as follows:...
-
On March 1, 2025, Blossom Construction Company contracted to construct a factory building for Bridgeport Manufacturing Inc. for a total contract price of $8,430,000. The building was completed by...
-
Moreover, we should oppose school voucher programs because any government program that lacks accountability and oversight should be prohibited. It is clear school voucher programs would lack such...
-
Ignoring double-bond stereo chemistry, what products would you expect from elimination reactions of the following alkyl halides? Which will be the major product in eachcase? Br CH CH CI CH (a) (b) Br...
-
What stereo chemistry do you expect for the alkene obtained by E2 elimination of (1R, 2R)-1, 2-dibromo-1, 2-diphenylethane? Draw a Newman projection of the reacting conformation?
-
The satellite DNAs of Drosophila virilis can be isolated, essentially free of main-fraction DNA, by density-gradient centrifugation. If these satellite DNAs are sheared into approximately...
-
The Clements want to start improving their financial situation in anticipation of buying their dream house. What effect would it have on their net worth and cash flows if they liquidated enough of...
-
City Farm Insurance has collection centers across the country to speed up collections. The company also makes its disbursements from remote disbursement centers so the firm's checks will take longer...
-
Explain stakeholder theory. How can stakeholder theory be reconciled with a theory that says a firm's sole purpose is to maximize shareholder wealth?
-
Explain the conflict resolution techniques with Examples
-
Crane Corporation produces wooden and aluminum baseball bats. In preparing the current budget, Crane's management estimated a total of $ 5 4 0 , 0 0 0 in manufacturing overhead costs and 1 8 , 0 0 0...
-
Using real-world examples, evaluate the effectiveness of foreign aid in promoting economic growth and economic development.
-
Draw the appropriate control flow graph of the given pseudocode.Make sure to only use one number for blocks of code which are all sequential and when the first line is executed, all of those lines...
-
Consider this reaction at equilibrium: Predict whether the reaction will shift left, shift right, or remain unchanged after each disturbance. a. O 2 is removed from the reaction mixture. b. KCl is...
-
Propose a mechanism for conversion of the dianion to the ketone under mildly acidic conditions.
-
Show how the following ketones might be synthesized from the indicated acids, using any necessary reagents. (a) Propiophenone from propionic acid (two ways, using alkylation of the acid and using...
-
Propose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanism begins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilic acyl substitution...
-
Recently, there has been a resurgence in labor union organization and a subsequent increase in union membership across the United States. What can today's labor unions learn from their past 140-year...
-
). Prove by contraposition: "Vm, ne Z, if m -n is even, then m - n is even (15 points)
-
The production function is Y = AKL-a (1) where L is labor at time t, K, is capital at time t, A, is TFP at time t and a (0, 1). The law of motion for capital is K = 8Y-8K Assume that TFP evolves...

Study smarter with the SolutionInn App


