What is the functional group present in these compounds? SCH3 SH C CH, - Ph b) )
Question:
What is the functional group present in these compounds?
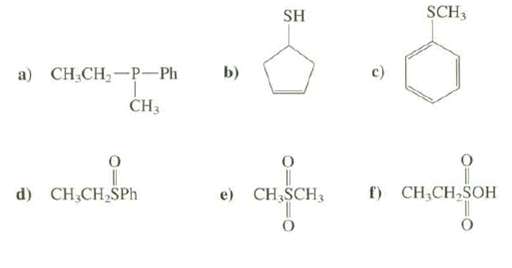
Transcribed Image Text:
SCH3 SH CН CH, - р—Ph b) а) ČH3 f) CH,CH,SOH d) CH,CH,SPh e) CH,SCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a Phosphine and arene ...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the functional difference between regular PCR and qPCCR?
-
What is the functional type of mannitol salt agar (MSA)? Explain how it is utilized in the isolation and identification of Staphylococcus species.
-
What is the functional definition of START? Cancer cells typically lose START control. Explain how the following mutations, which arc found in some cancer cells, lead to a bypass of START controls:...
-
Required information [The following information applies to the questions displayed below.] Most Company has an opportunity to invest in one of two new projects. Project Y requires a $310,000...
-
Doug Klock, 56, just retired after 31 years of teaching. He is a husband and father of three children, two whom are still dependent. He received a 141,000 lump-sum retirement bonus and will receive...
-
Describe the customer loyalty model.
-
After rereading the Opening Profile, identify all effective actions Ellie Symes and Wyatt Wells initiated in the early days of building The Bee Corp.
-
As you can see in the following table, demand for heart transplant surgery at Washington General Hospital has increased steadily in the past few years: The director of medical services predicted 6...
-
(a) A company has an EPS of Rs. 2.5 for the last year and the DPS of Rs. 1. The earnings is expected to grow at 2% a year in long run. Currently it is trading at 7 times its earnings. If the required...
-
Colson went to Logon Drugs and bought some light bulbs for the hallways of his new apartment building. A tenant asked for one and Colson gave him one but told him he was responsible for getting his...
-
Explain which compound has the higher melting point or boiling point: a) Melting point b) Boiling point or C COCH c) Boiling point or
-
Identify the most acidic site in thesecompounds: NH2 CH.COCH,CH,CH3 c) a) b) CH3 e) CH;CH,CH,COH d) CH,CH,CCH,CH3
-
Access the latest annual report for Starbucks Corporation by clicking on the Investor Relations and Annual Reports links at www.starbucks.com. Required Review the first note to the financial...
-
Before buying a car, your aunt spent months choosing between two options. after she decided what car to buy, she claimed that the right choice was always obvious because the one car is so much...
-
1. Two techniques that are often used to financially evaluate capital projects include IRR and NPV. Describe which you believe is preferable and why. A discussion of disadvantages of either approach...
-
Simplify the following completely 9x3 (2x-2)+3x2(-9x-6) 3x2 Answer = Question Help: Message instructor
-
Consider the problem u' (t): = ku, u(0) = 1 for constant k and t > 0. (a) Find an explicit formula in terms of h, k, and i for the Euler solution u, at t = ih. (b) Find values of k and h such that |u...
-
How truly does credit and stock administration impact corporate money choices for a situation study, and what methodologies can be utilized to streamline an organization's credit and stock practices?
-
The revenue and related profit under an Istisnaa contract are recorded in each financial period using: a. Percentage of completion method only b. Cost to complete method only c. Percentage of...
-
Could a set of three vectors in span all of? Explain. What about n vectors in when n is less than m? R4
-
The classic example of monopsony is the company town a small community in which a single firm is the dominant employer. In extreme examples, companies have owned and managed all housing, stores, and...
-
What sequence of bases on one strand of DNA is complementary to the following sequence on another strand? (5') GGCTAATCCGT (3')
-
Show how uracil can form strong hydrogen bonds to adenine.
-
What RNA base sequence is complementary to the following DNA base sequence? (5') GATTACCGTA (3')
-
a) Give examples of low, medium and high strain rate test types. [4 marks] b) A plot of log (stress) against log (strain rate) provides a straight line relationship. Based on this, provide an...
-
3- Let the density of the pyramid material be p = 2000 kg/m. Determine the average normal stress at a cross section located at x measured from the apex. Express the stress as a function of x. Assume...
-
You are designing a wing with no aerodynamic twist based on the airfoil camber given by: N C 3.1 (H-H) = 0.1 The wing will have an aspect ratio of 10, a taper ratio of 0.8, and a constant geometric...

Study smarter with the SolutionInn App


