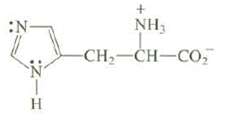
What is the hybridization at each nitrogen of the amino acid histidine? What kind of orbital is
Question:
What is the hybridization at each nitrogen of the amino acid histidine? What kind of orbital is occupied by the unshared pair of electrons on each nitrogen? Explain.
Transcribed Image Text:
:N :Z-H H + NH₂ CH, CH–CO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Nitrogen 1 is sp hybridized and its unshared pair of electrons i...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the hybridization at each C in this molecule? Indicate the type of bond and the orbital's that are overlapping to form it for each of the designated bonds? ITT H=C=C=C=C_C7H (both) H tall...
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
What is the hybridization at all atoms, except hydrogen's in these compounds? a) CHNH, d) b) CH=CHCHC=N OH 6 H NH
-
A dipole of 6.6 nC point charges arranged 0.16 cm apart is centered at the origin and oriented along the x-axis. The positive charge is on the negative x axis. How m energy is required to place a...
-
Broome Instruments Company manufactures a control valve used in air-conditioning systems. The firm uses a standard costing system for product costing. The manufacturing overhead rate is based on a...
-
Jack and Jill's Place is a nonprofit nursery school run by the parents of the enrolled children. Since the school is out of town, it has a well rather than a city water supply. Lately, the well has...
-
Identify requirement categories.
-
Information concerning Montana Company is provided in BE19-16. What are the total product costs for the company under absorption costing?
-
Upsidedown Cake Company produces dessert products for sale in grocery stores, but it also has a retail location. At the end of 2 0 2 3 , the company had $ 3 89 , 0 0 0 in accounts receivable before...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
Show a MO energy level diagram for the neutral molecule HeH. Use this diagram to explain whether HeH is expected to stable or not?
-
At a pH of 10.8, the amino acid arginine exists primarily as the following dipolar ion. Show the resonance structures for the cationic part of arginine and discuss their relative contributions to the...
-
On December 31, 2016, Eli Associates owned the following long-term investments. Ordinary Shares Shares Cost Trowbridge Co....................4,000...............96,000 Holly...
-
Graph the lines in Problems 31-44. \(y=2 x+3\)
-
For all nonhomicide offenses, there are five general elements of duress: (1) The defendant acted under the compulsion or threat of imminent infliction of death or great bodily injury; (2) the...
-
Don kidnapped his seven-year-old daughter, Brittany, after the family court refused to give him legal visitation rights. Don and Brittany are on the run and need a place to hide from authorities. Don...
-
There are three categories of creative excuse defenses: excuses based on physiology, excuses based on psychology, and sociological excuses. Examples of creative physiological excuse defenses include...
-
The insanity defense is distinct from competency to stand trial. The latter deals with the defendants ability to understand what is happening at trial (as well as at pretrial hearings, etc.). ...
-
In June 2006, the Commodities Futures Trading Commission filed a civil complaint against \(\mathrm{BP}\), alleging that its brokers tried to manipulate the price of propane by manipulating the...
-
True & False The basis of an asset must be reduced by the depreciation allowable, 2. Adjusted gross income (AGI) is the basis for a number of phase-outs of deductions. 3. A change to adjusted gross...
-
Test the series for convergence or divergence. (-1)+1, M=1 n? 3 n' + 1
-
Draw the two chair conformations of menthol, and tell which is morestable. C Menthol CH(CH3)2
-
There are four cistransisomers of menthol (Problem 4.37), including the one shown. Draw the other three.
-
Identify each pair of relationships among the ? OH groups in glucose (red?blue, red?green, red?black, blue?green, blue?black, green?black) as cis or trans. CH2OH OH OH Glucose OH
-
What are the advantages and disadvantages of utilizing PostgreSQL's advanced indexing techniques such as GiST and GIN?
-
discuss the implications and trade-offs of utilizing PostgreSQL's advanced security features such as row-level security (RLS) and column-level encryption in compliance-sensitive applications...
-
How does PostgreSQL's support for advanced full-text search capabilities through extensions like pg_trgm and tsearch improve search functionality and performance in applications requiring...

Study smarter with the SolutionInn App


