What product would expect to obtain from base treatment of 1,6-cyclo-decacedione? Base 1,6-Cyclodecanedione
Question:
What product would expect to obtain from base treatment of 1,6-cyclo-decacedione?
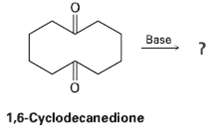
Transcribed Image Text:
Base 1,6-Cyclodecanedione
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
This intramolecular aldol con...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What product would you expect to obtain from the base-catalyzed Michael reaction of (a) 1, 3-diphenylprop-2-en-1-one (Section 19.5A) and acetophenone (b) 1, 3-diphenylprop-2-en-1-one and...
-
What product would you expect to obtain from each of the following reactions? a. b. c. CH3CH2CHCH CH2CH2COH CH COCH CH HCI CH OH CH CH COH 1. SOC2 2. AICI3 3. H20
-
What product would you expect to obtain from each of the following reactions? (a) (b) NaNO2, HCI SO2Cl aq. KOH
-
Bilge Pumpworks and Seaworthy Rope Company agreed to merge on January 1, 20X3. On the date of the merger agreement, the companies reported the following data: Bilge Pumpworks has 10,000 shares of its...
-
Major professional schools are highly competitive and most applicants do not get past the admissions process. That makes admissions a critical gatekeeper function for these schools. Given that, why...
-
Formic acid, HCOOH, is a weak electrolyte. What solute particles are present in an aqueous solution of this compound? Write the chemical equation for the ionization of HCOOH.
-
Why is data so critical to the development of machine learning applications? How is data used by machine learning developers?
-
Olongapo Sports Corporation distributes two premium golf ballsthe Flight Dynamic and the Sure Shot. Monthly sales and the contribution margin ratios for the two products follow: Fixed expenses total...
-
How do organizational culture and climate influence employee engagement, job satisfaction, and retention rates, particularly in multinational corporations operating in diverse cultural contexts?
-
Exhibit 1.25 presents common-size income statements and balance sheets for seven firms that operate at various stages in the value chain for the pharmaceutical industry. These common-size statements...
-
Treatments of a 1, 3-diketone such as 2,4-pentanedione with base does not give an aldol condensation product. Explain.
-
Show the products you would except to obtain by claisen condensation of the following esters: (a) (CH3)2CHCH2COEt (b) Ethyl phenyl acetate (c) Ethyl cyclohexylacetate
-
Phillips Co. is growing quickly. Dividends are expected to grow at a rate of 25 percent for the next three years, with the growth rate falling off to a constant 5 percent thereafter. If the required...
-
A married couple, Simon and Maria, seek advice from you on their retirement life. Simon is 65 years old and Maria is 62, and they wish to retire on a comfortable amount. Simon's life expectancy at...
-
A company is considering investing $1.5 million in a new production system today (year 0). The new production system will result in an after-tax cost savings of $200,000 at the end of year 1. These...
-
The project is expected to cost $4 million and generate after-tax net cash flows of $200,000 at the end of year 1, $300,000 at the end of year 2, $400,000 at the end of year 3. The after-tax net cash...
-
Mr A has been working for several years as a senior manager in France for the company AVIRON. A year ago, his employer offered him to take over the technical management of its subsidiary AVIRON ON...
-
What is child abuse? Most prevalent? Types? Historical discoveries of abuse? Most prevalent? The relationship between culture and child abuse i.e. Indigenous Factors associated with causes of child...
-
A salad spinner (Figure P11.11) is used to dry lettuce that has been washed. What physics is going on as the spinner does its job? Data from Figure P11.11
-
In each of the following independent cases, document the system using whatever technique(s) your instructor specifies. a. Dreambox Creations (www.dreamboxcreations.com/) in Diamond Bar, California,...
-
Diazomethane is a highly poisonous, explosive compound because it readily evolves N 2 . Diazomethane has the following composition by mass: 28.57% C; 4.80% H; and 66.64% N. The molar mass of...
-
Give the structures of the following compounds. (a) Methylene iodide (b) Carbon tetrabromide (c) 3-bromo-2-methylpentane (d) Iodoform (e) 2-bromo-3-ethyl-2-methylhexane (f) Isobutyl bromide (g)...
-
Draw a perspective structure or a Fischer projection for the products of the following SN2 reactions. (a) trans-1-bromo-3-methylcyclopentane + KOH (b) (R)-2-bromopentane + KCN (c) (d) (e) (f) CH Br-H...
-
Under appropriate conditions, (S)-1-bromo-1-fluoroethane reacts with sodium methoxide to give pure (S)-1-fluoro-1-methoxyethane. (a) Why is bromide rather than fluoride replaced? (b) Draw perspective...
-
Melissa Cutt is thinking about buying some shares of EZLawn Equipment, at $36.44 per share. She expects the price of the stock to rise to $43.62 over the next 3 years. During that time she also...
-
At the beginning of the week, Taylan's restaurant had a beginning inventory of food and beverages totaling $4,000. During the week, additional food and beverages purchases totaled $3,000. At the end...
-
1. Identify the auditor's role in the internal and external auditing processes. 2. Identify one law, regulation, and statue affecting healthcare

Study smarter with the SolutionInn App


