What products would you expect from Hofmann elimination of the following amines? If more than one product
Question:
What products would you expect from Hofmann elimination of the following amines? If more than one product is formed, indicate which ismajor.
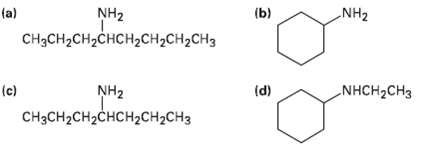
Transcribed Image Text:
NH2 (b) NH2 (a) CH3CH2CH2CHCH2CH2CH2CH3 NHCH2CH3 (d) NH2 (c) CнзCH2CH2сHCHасH2CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
The Hofmann elimination yields alkenes and amines from larger amines The major alkene ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What product would you expect from Hofmann elimination of a heterocyclic amine such as piperidine? Write all thesteps? Piperidine
-
What products would you expect from each of the following reactions? (a) (b) (c) t-BuOK CHBr CH2Zn(Cu) diethyl ether
-
What products would you expect to obtain when ethyl propanoate reacts with each of the following? (a) H3O+, H2O (b) HO-, H2O (c) 1-Octanol, HCl (d) CH3NH2 (e) LiAlH4, then H2O (f) Excess C6H5MgBr,...
-
How can adults continue to function relatively normally after surgery to remove the thymus, tonsils, spleen, or lymph nodes?
-
IF an employer's hiring policies give preference to those who are already employed, what is the impact on (a) the company's performance; (b) workers seeking jobs: (c) the communities where a company...
-
Wolfsburg Wagon (WW) is a small automaker. The accompanying table shows WWs long run average total cost. Quantity of cars LRATC of car 1 $30,000 2 20,000 3 15,000 4 12,000 5 12,000 6 12,000 7 14,000...
-
In Example 6.7, let \(m_{1}=3 m_{2}\). (a) Where on axis A is the center of mass of the two-cart system? (b) Where on axis A would you need to place a third cart of inertia \(m_{3}=m_{1}\) so that...
-
Bill Watts, president of Western Publications, accepts a capital budgeting project proposed by Division X. this is the division in which the president spent his first 10 years with the company. ON...
-
The amount of realized gain to be recognized in a tax year is equal to the gross profit multiplied by the ratio of payments received in the current year divided by the total contract price. ...
-
Cary Company manufactures two models of industrial componentsa Standard model and an Advanced Model. It has provided the following information with respect to these two products: Standard Advanced...
-
How would you prepare the following amines, using both Hofmann and Curtius rearrangements on a carboxylic acidderivative? CH (b) (a) NH2 CH2CH2NH2 CH
-
Propose a synthesis of the drug sulfathiazole from benzene and any necessaryamine. Sulfathiazole H2N
-
Joe Fat cat, an investment banker, states: It is not worth my while to worry about detailed, long-term forecasts. Instead, I use the following approach when forecasting cash flows beyond three years....
-
Describe the four different components of a time-series statistics.
-
What is multicollinearity in a regression model?
-
List ten questions you would ask a group of passengers in order to estimate their demand function for a specific airline and a specific route.
-
In a simple regression model, is it possible that all the actual Y values would lie above or below the true regression line? Explain.
-
Write the demand equation, in general form, for an airline and identify the following terms: a. Dependent variable b. Independent variables c. Y-intercept d. Slope e. Logically, one can often expect...
-
Gamma Petroleum owns 100% of the working interest in the Bearcat Field in Wyoming. The field has been producing for 3 years and is expected to produce for 10 more years. Since the state has very...
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
The equilibrium constant for the reaction SO 2 (g) + NO 2 (g) SO 3 (g) + NO(g) is Kc = 3.0. Find the amount of NO 2 that must be added to 2.4 mol of SO 2 in order to form 1.2 mol of SO3 at...
-
Examine the three ball-and-stick models shown below: (a) Redraw the three structures using solid lines, dashed wedges, and solid wedges. (b)What is the relationship, identical or isomers, between...
-
Write a structural formula that corresponds to the molecular formula C3H6O and is a. Acyclic b. Carbocyclic c. Heterocyclic
-
Draw a chemical structure for diacetyl (2,3-butanedione), a diketone with the C4H6O2 chemical formula. Diacetyl is used as a flavoring for microwave popcorn, but has been under scrutiny of late as a...
-
O O A Dundas Company's inventory records for its retail division show the following at May 31: (Click the icon to view the accounting records.) At May 31, 10 of these units are on hand. Dundas...
-
4. R got 35% hike in his salary and 20% incentive on sales. If R sold goods worth Rs. 850 last year and the salary was Rs 70, then how much more does he earn this year with sales of Rs.900?
-
Teachers salaries have not been increasing as fast as the pay in many other types of work. Explain why you think this might be true. use graph of the supply and demand for teacher's labor to help...

Study smarter with the SolutionInn App


