Where in the IR spectrum would you expect each of the following molecules toabsorb? (a) (c) (b)
Question:
Where in the IR spectrum would you expect each of the following molecules toabsorb?
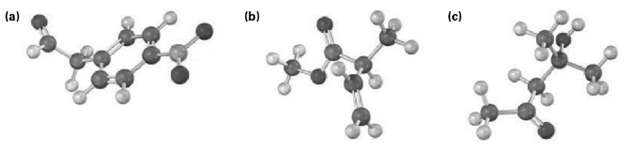
Transcribed Image Text:
(a) (c) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
a b c Compound H HC 0 CH3 3 CH 2 HH H3COH 12 CH3 Significa...View the full answer

Answered By

Benard Ndini Mwendwa
I am a graduate from Kenya. I managed to score one of the highest levels in my BS. I have experience in academic writing since I have been working as a freelancer in most of my time. I am willing to help other students attain better grades in their academic portfolio. Thank you.
4.90+
107+ Reviews
240+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In which direction would you expect each of the following equilibria to lie? (a) NH 3 + - OH NH 2 - + H 2 O (b) CH 3 NH 2 + H 2 O CH 3 NH 3 + + - OH (c) CH 3 NH 2 + (CH 3 ) 3 NH + CH 3 NH 3 + +...
-
Would you expect each of the following atoms to gain or lose electrons when forming ions? What ion is the most likely in each case? a. Ra b. In c. P d. Te e. Br f. Rb
-
How many signals would you expect each of the following molecules to have in its 1H and 13Cspectra? (c) CH -H (b) () CH CHCH CH C CH (f) (e) (d) "T CH "
-
Since perpetuity payments continue forever, how can a present value be computed? Why isnt the present value infinite?
-
"Niche strategies are generally more profitable than "mass-market" strategies because they usually imply weaker price competition." Comment.
-
For the circuit in Fig. 7.113, is(t) = 5u(t) Find v(t). 2 6 0.25 F
-
For laminar free convection flow on a vertical plate, the recommended values of \(C\) and \(n\) for use in the correlation of Equation 9.24 are 0.59 and 1/4, respectively. Derive the values of \(C\)...
-
On January 1, 2017, Kimbel Inc. had these stockholders' equity balances Common Stock, $1 par (2,000,000 shares authorized, 600,000 shares issued and outstanding) .............................$...
-
1. $3,800+ ($8,000) =$ (-4,200) Netting of long term capital gains and losses ($2,400) + 2,000 + ($500) = $ (-900) Netting of short term capital gains and losses (-4,200) + (-900) = $(-5,100) Netting...
-
Write Significance of social study at elementary level...(in detail)
-
Where might the following compound have IR absorptions?
-
Show the structures of the likely fragments you would expect in the mass spectra of the followingmolecules: (a) (b)
-
Convert the quantity to the indicated units. When appropriate, round your answer to the nearest hundredth. 6.2 acres to hectares
-
Are the statements made by Robert F Kennedy accurate? What are some of the major issues with using GDP as a measure of economic well-being? What specifically do you think can be done to come up with...
-
How much is the EPS growth for droneshield company (ASX: DRO) and how to calculate it?
-
N23 B 1 Function: RATE, Formula: Multiply, Subtract; Cell Referencing 2 3 Using Excel to Account for Notes Payable 4 PROBLEM X fx 9 10 5 Snade Corporation issued a five-year note to Hames Corp. on...
-
Use the one-period model to explain why a tax on capital is not a good idea. Determine the effects of capital tax on aggregate output, consumption, employment, and the real wage. Please use diagrams...
-
A company wants to promote a healthy lifestyle and therefore offers its employees a discount on the employee-paid portion of their health insurance premiums if they participate in a health and...
-
Using the approach in Section 4.4, find the controllable canonical form for the plant transfer function and then design a full-state feedback controller that places the closed-loop poles at the same...
-
Suppose that you are part of a virtual team and must persuade other team members on an important matter (such as switching suppliers or altering the project deadline). Assuming that you cannot visit...
-
Substance A is a nonpolar liquid and has only dispersion forces among its constituent particles. Substance B is also a nonpolar liquid and has about the same magnitude of dispersion forces among its...
-
The fragrance of (Z)-1-phenylhex-2-en-1-ol resembles that of roses, with a delicate citrus edge. Show how you would synthesize this compound from benzaldehyde (PhCHO) and any other reagents you need.
-
In the addition of just 1 mole of bromine to 1 mole of hex-1-yne, should the hex-1-yne be added to a bromine solution or should the bromine be added to the hex-1-yne? Explain your answer.
-
Propose a mechanism for the entire reaction of pent-1-yne with 2 moles of HBr. Show why Markovnikov's rule should be observed in both the first and second additions of HBr.
-
The Production Department of Hruska Corporation has submitted the following forecast of units to be produced by quarter for the upcoming fiscal year: Units to be produced 1st Quarter 2nd Quarter 3rd...
-
You go to the hardware store to buy a new 50 ft garden hose. You find you can choose between hoses of inch and inch inner diameter. Compare the rate at which water flows through the two types of...
-
Miller Company's contribution format income statement for the most recent month is shown below: Sales (32,000 units) Variable expenses Total $ 224,000 128,000 Contribution margin 96,000 Per Unit $...

Study smarter with the SolutionInn App


