Which of the following compounds can probably be prepared by a mixed aldol reaction? Show the reactants
Question:
Which of the following compounds can probably be prepared by a mixed aldol reaction? Show the reactants you would use in eachcase.
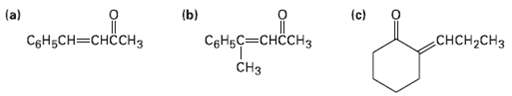
Transcribed Image Text:
смсн- (b) (a) (c) сnрове СоНьСн—снсснз снсH-CHз CеHьС— снҫсHз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
a CH O CCH3 NaOH i CH3CCH3 CH3CCH3 EtOH 4Phenyl3buten2one This mixed aldol will succe...View the full answer

Answered By

Sarah Khan
My core expertise are:
-_ Finance
-_ Business
-_ Management
-_ Marketing Management
-_ Financial Management
-_ Corporate Finance
-_ HRM etc...
I have 7+ years of experience as an online tutor. I have hands-on experience in handling:
-_ Academic Papers
-_ Research Paper
-_ Dissertation Paper
-_ Case study analysis
-_ Research Proposals
-_ Business Plan
-_ Complexed financial calculations in excel
-_ Home Work Assistance
-_ PPT
-_ Thesis Paper
-_ Capstone Papers
-_ Essay Writing etc...
5.00+
91+ Reviews
92+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following compounds can exist as cis-trans isomers? Draw their structures. a. Pentene b. 3-heptene c. 2-methyl-2-pentene d. 2-hexene
-
Which of the following compounds can exist as cis-trans isomers? If such isomerism is possible, draw the structures in a way that clearly illustrates the geometry. a. 3-octene b. 3-chloropropene c....
-
Show how each of the following compounds can be synthesized from cyclopentanol and any necessary organic or inorganic reagents. In many cases the desired compound can be made from one prepared in an...
-
Assume that on July 1, 2014, Big Corp. loaned little Corp. $12,000 for aperiod of one year at 6percent interest. What amount of interest revenue will big report for 2014? What amount of cash will big...
-
How would you go about identifying the powerful people within your organization? What indicators would you look for? From what types of problems would these indicators suffer?
-
(a) A strontium hydroxide solution is prepared by dissolving 10.45 g of Sr(OH)2 in water to make 50.00 mL of solution. What is the molarity of this solution? (b) Next the strontium hydroxide solution...
-
The development stages of artificial intelligence are described as weak, strong, and super intelligent AI. Explain how applications in each stage will differ from those in other stages.
-
Haley Company, organized in 2014, has these transactions related to intangible assets in that year: Jan. 2Purchased a patent (5-year life) $280,000. Apr. 1 Goodwill acquired as a result of purchased...
-
Cite three ways that global HR differs from domestic HR, specifically in relation to Total Rewards. Give examples, and cite at least one source from the class. Respond to at least one colleague.?
-
On September 2, 2010, the Securities & Exchange Commission brought an action against Sujata Sachdeva, vice president of finance, and Koss senior accountant and subordinate, Julie Mulvaney, who...
-
Show how you would synthesize the following compound using an aldolreaction:
-
Treatments of a 1, 3-diketone such as 2,4-pentanedione with base does not give an aldol condensation product. Explain.
-
A curve is such that d 2 y/dx 2 = 8x. The curve has a maximum point when x = 1, and the point (2, 1) lies on the curve. Find the equation of the curve.
-
Suppose you invest $1,000 today, compounded quarterly, with the annual interest rate of 5%. What is your investment worth in one year?
-
The yield on the company's outstanding bonds is 7.75%, its tax rate is 25%, the next expected dividend is $0.65 a share, the dividend is expected to grow at a constant rate of 6.00% a year, the price...
-
A treasurer of an Australian bank issues a 1-year, AUD$1 million CD at 2.7 percent annual interest to finance a Canadian investment. The treasurer invests C $1.307 million in 2-year fixed-rate...
-
Monash bank has 4-year zero-coupon bonds with a total fair value of $27 million. The current market yield on the bonds is 6.7 percent. The maximum potential adverse move in yields is estimated at 19...
-
A firm has sold a product in Switzerland with a payment of SFr 4,000,000 to be received in three months. The following market quotes are available: firm-specific investing iUS: 6.00% per year firm...
-
The Ferris wheel in Figure P11.9 is turning at constant speed. Draw and label free-body diagrams showing the forces on passengers at the numbered positions. Be careful to show correct relative...
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
NaCl has a lattice energy of -787 kJ/mol. Consider a hypothetical salt XY. X 3 + has the same radius as Na + and Y 3 has the same radius as Cl . Estimate the lattice energy of XY.
-
Predict the major products of the following substitutions. (a) (b) (c) (d) (e) (f) CH3CH2B(CH3)3CO ethyl bromide potassium tert-butoxide sodium acetylide 1-chlorobutane CH32CHCH2Br + excess NH3 CH...
-
Show how you might use SN2 reactions to convert 1-chlorobutane into the following compounds. (a) Butan-1-ol (b) 1-fluorobutane (c) 1-iodobutane (d) CH3-(CH2)3-CN (e) (f) CH3CH2-O-(CH2)3-CH3 (g) CH3-...
-
For each pair, predict the stronger nucleophile in the SN2 reaction (using an alcohol as the solvent). Explain your prediction. (a) CH3CH2)3N or (CH3CH2)2NH (b) (CH3)2O or (CH3)2S (c) NH3 or PH3 (d)...
-
A car trip lasts 3h 36 min at 86 km/h, including a half-hour break for lunch. How much time would we save by driving at 100 km/h without taking a break? (When the automobile is moving, we assume that...
-
Sylvia is interested in financial laws and regulations. She enjo make sure they are in compliance with laws and regulations. career should Sylvia consider?
-
Mariah Lucas is restructuring the budget process at her company, Lucas Lodging, Inc. She envisions a budget process driven by management and lower-level employees, but aligned with the company's...

Study smarter with the SolutionInn App


