Which of the following Fischer projections of glyceraldehydes represent the sameenantiomer? CH- -
Question:
Which of the following Fischer projections of glyceraldehydes represent the sameenantiomer?
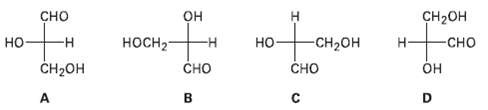
Transcribed Image Text:
CH-он он сно -н Н но носн3 -н -CH-он Н- -сно но он CH2он A Сно Сно
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
To decide if two Fischer projections are identical use the two allowa...View the full answer

Answered By

Navashree Ghosh
I believe in quality work and customer satisfaction. So, I can assure you that you will get quality work from me when you hire me. Let's work together and build a long-term association.
4.90+
82+ Reviews
116+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Convert the following Fischer projections into tetrahedral representations, and assign R or S stereochemistry toeach: H (b) (a) H2N- - (c) - - CH-CH CH CH
-
Which of the following Fischer projection formulas have the same configuration as A, and which are the enantiomer of A? C-H, HO-CH C2H5 b. CHs c. HCH3 . a. H-OH C2Hs
-
Convert the following Fischer projections to perspective formulas. (a) (b) (c) (d) H,N- H- CH;
-
When a manager greets department visitors or attends ceremonies, he or she is playing the role of: Figurehead Conceptual Empowerment Productivity
-
You know those times when you're craving Thai food or the perfect fruit smoothie, but you don't know where to go? Or when you're out shopping or clubbing and want to let your friends know where you...
-
A cylindrical metal wire 2 mm (0.08 in.) in diameter is required to carry a current of 10 A with a minimum of 0.03 V drop per foot (300 mm) of wire. Which of the metals and alloys listed in Table...
-
List five topics which are usually referred to in audit engagement letters.
-
Minsoo Ltd. is a retailer operating in Edmonton, Alberta. Minsoo uses the perpetual inventory method. All sales returns from customers result in the goods being returned to inventory; the inventory...
-
Question One You must answer all parts of this question Jane runs a meeting group for parents with babies and toddlers, the "Group for Babies and Toddlers" (GBT) in the town of Glead, Ireland. She...
-
1. Are the auditors guilty of malpractice? Explain. 2. What defenses are available to Joker & Wild in this case? Explain what they must prove to successfully assert these defenses. 3. Assume you are...
-
Classify each of the following monosaccharide?s: la) 0 (c) (b) (d) CH- . H- C=0 C=0 -- -- -- -- -- -- -- -- -- H2OH -- CH- Ribulose CH Threose CH- Tagatose 2-Deoxyribose
-
Redraw the following molecule as a Fischer projection, and assign R or S configuration to the chirality center (yellow-green =Cl):
-
The fermentation of corn produces a mixture of 3.3 mol% ethyl alcohol in water. If this mixture is distilled at 1 atm by a differential distillation, calculate and plot the instantaneous-vapor...
-
TCSS 143 A Programming Assignment 2 Due: May 13th 2023, 11:59 PM on Canvas NOTE: Be sure to adhere to the University's Policy on Academic Integrity. Programming assignments are to be written...
-
Barrington Inc. has never paid dividends before. Today, it made a public announcement that it will pay an annual dividend of $2.00 two year from now. It also stated that dividends would increase by...
-
F is an all-equity firm with assets worth $1Mns. Expected returns on assets equal to 9%. F buys new assets at market value for $0.5Mns (same risk as existing assets). The acquisition is financed by...
-
Beginning Work-in-Process Inventory Direct Materials Used Direct Labor Manufacturing Overhead Total Manufacturing Costs Incurred during the Year Total Manufacturing Costs to Account For Ending...
-
How much must you invest today at 10% interest in order to see your investment grow to $8,000 in 3 years? To save for a new car, Samuel will invest $18,000 at the end of each year for the next 5...
-
For your twenty-first birthday, your grandfather offers you a gift of \($1\),000 today. However, you have the choice of waiting 3 years and receiving \($1\),500 or waiting 5 years and receiving...
-
Pedro Bourbone is the founder and owner of a highly successful small business and, over the past several years, has accumulated a significant amount of personal wealth. His portfolio of stocks and...
-
Find the pH of 0.175 M NaCN solution. For HCN, K a = 4.9 * 10 -10 . a) 5.03 b) 11.28 c) 2.31 d) 8.97
-
As discussed in "A Word About . . . Vitamin E-Tocopherols and Tocotrienols" on page 128, these compounds can be obtained from different sources. How would you convert a-tocotrienol to a-tocopherol?
-
For the compounds named below, (1) draw the structure of each compound. (2) using benzene or toluene as the only aromatic starting material, devise a synthesis of each compound. a. P-bromotoluene b....
-
Using benzene or toluene as the only aromatic organic starting material, devise a synthesis for each of the following compounds. Name the product. CH CH3 Cl CH3 Cl a. b. C. d. Br NO2 NO2
-
Bailey Delivery Company, Incorporated, was organized in 2021 in Wisconsin. The following transactions occurred during the year: a. Received cash from investors in exchange for 17,000 shares of stock...
-
Write a program (C++) that prompts the user to input an integer and then outputs both the individual digits of the number and the sum of the digits. Example: Input: 3456 Output: 3 4 5 6 Sum of digits...
-
The soil profile at a site consists of 1.5 m of sand underlain by a 6 m thick clay layer. The water table coincides with the top of the sand layer. The saturated unit weight (Ysat) of the sand and...

Study smarter with the SolutionInn App


