Which of these compounds exhibit cis-trans isomerism? Draw both cis-trans isomers when they exist? a) CHCHCH=CHCH CH3
Question:
Which of these compounds exhibit cis-trans isomerism? Draw both cis-trans isomers when they exist?
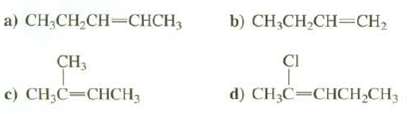
Transcribed Image Text:
a) CH₂CH₂CH=CHCH₂ CH3 c) CH₂C=CHCH₂ b) CH3CH₂CH=CH₂ CI d) CH₂C=CHCH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
Cistrans isomers are stereo isomers that differ in the placement of groups about ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
Which of these compounds can be a member of an isomer pair? In each case where isomerism is possible, identify the type or types of isomerism. [Sections 24.2, 24.4] CH2 C C-OH --O NHE Cl (b) CH3CH2CH...
-
Draw the cis and trans isomers for the following compounds: a. 1-ethyl-3-methylcyclobutane b. 2-methyl-3-heptene c. 1-bromo-4-chlorocyclohexane d. 1,3-dibromocyclobutane
-
What Codification citation indicates how unrealized gains or losses from changes in fair value affect net income when investments are classified as available-for-sale?
-
Distinguish between a support department and a production department. Give an example of a production department in a bank, and a support department in a restaurant chain.
-
a. Are items of income not listed in Sec. 61 taxable? Explain. b. Because there is no specific exclusion for unrealized income, why is it not taxable? c. Can income be realized even when a...
-
Consider \(20{ }^{\circ} \mathrm{C}\) water flowing over a thin, wide, smooth flat plate aligned with the flow. The approach velocity is \(60 \mathrm{~km} / \mathrm{hr}\) and the Reynolds number...
-
Assume you work at a regional state university whose traditional target market, high school students within your region, is shrinking and projections are that this segment will decrease approximately...
-
Mumford Limited had the following financial statement account balances for 2022. Net Sales $356,420 Cost of Goods Sold 152,000 Average Accounts Receivable 19,710 Average Inventory 4,845 average...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
Explain which compound has the higher solubility in water.
-
Draw the cis-trans isomers for these compounds and explain which is more stable: CH3 a) CH CH CHCOH NH c) e CH, CH2NHCH CH f CH3CH CH CH
-
A company accepts user orders for its products interactively. Users might make the following errors as they enter data: The item number ordered is not numeric, too low (less than 0), or too high...
-
In analysis, some focus seems to be on the need for NPV equations to be applied to projects that are mutually exclusive. But in practice we find that the lines are blurred in capital budgeting....
-
The market value of a Venturi pump averages $\$ 195.50$. There are a large number of suppliers of the pump, and prices vary only slightly from the mean, but are still normally distributed. If the...
-
In developing a capital budgeting portfolio, there are times when a project with a lower return is squeezed in to the budget but a higher-return project is excluded. How can this be fair? Wouldn't...
-
Consider a binary mixture of ideal gases, a and b, at temperature T and pressure P. Come up with an expression for in terms of T, P, and ya. What is the value of (AGmix) a
-
It was Saturday evening and Joe Johnson, VP of Patient Services, was relaxing with his family after dinner and watching a situation comedy. As the administrator on call (AOC), he was grateful things...
-
On May 7, Roy, a minor, a resident of Smithton, purchased an automobile from Royal Motors, Inc., for \($12,750\) in cash. On the same day, he bought a motor scooter from Marks, also a minor, for...
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
The region bounded by the curves y = arctan x, y = 0, and x = 1 is rotated about the y-axis. Find the volume of the resulting solid.
-
(a) How many degrees of unsaturation does a-terpinene have? (b) How many double bonds and how many rings does it have? (c) Propose a structure fora-terpinene. CCH-CH2CCHCH3 C-C H ...
-
Evidence that cleavage of 1, 2-diols by HIO 4 occurs through a five-membered cyclic periodate intermediate is based on kinetic data?the measurement of reaction rates. When diols A and B were prepared...
-
Reaction of HBr with 3-methylcyclohexene yields a mixture of four products: cis- and trans-1-bromo-3-methylcyclohexane and cis- and trans-1-bromo- 2-methylcyclohexane. The analogous reaction of HBr...
-
The imposition of a ceiling on the length of time a criminal case should take to get to trial is a multifaceted issue, balancing the defendant's right to a timely trial with the complexities of...
-
Discuss three changes written into the Constitution in 1789 that changed the government from what it was under the ArticIes of Confederation?
-
Assume South Park is initially closed to trade. Domestic supply and demand for winter hats is given by the following equations: D = 60 - 4P, S = 2P - 6. a. Suppose South Park opens up to trade and...

Study smarter with the SolutionInn App


