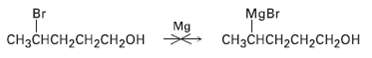
Why do you suppose it?s not possible to prepare a Grignard reagent from a bromo alcohol such
Question:
Why do you suppose it?s not possible to prepare a Grignard reagent from a bromo alcohol such as 4-bromo-1-pentanol? Give another example of a molecule that is unlikely to form a Grignard reagent.
Transcribed Image Text:
MgBr Br Mg CHнзснсH2CH2CH2он снзснсн2сH2сH2оH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
A Grignard reagent cant be prepared from a compound containing an acidic ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give another example of adverse selection.
-
Why do you suppose that the United Nations gives high, medium, and low projections for future population growth? Why not give a single estimate? What factors would you consider in making these...
-
Why do you suppose that forensic DNA analysis relies principally on short tandem repeats (repeat polymorphisms), rather than single- nucleotide polymorphisms, such as that described on page 669 and...
-
The director of cost management for Odessa Company uses a statistical control chart to help management determine when to investigate variances. The critical value is 1 standard deviation. The company...
-
Refer to the Flexible Insurance Coverage case. Design a spreadsheet that will allow an individual employee with estimated annual medical expenses of $400 to compare the total expenses under each...
-
(a) In what fundamental way is mass spectrometry related to Thomson's cathode-ray experiments (Figure 2.4)? (b) What are the labels on the axes of a mass spectrum? (c) To measure the mass spectrum of...
-
What is the purpose of an EDI procurement system?
-
Crable and Tesch, the systems consultants in P9-3, budgeted overhead and other expenses as follows for the year ended December 31, 2016: Overhead: Depreciationequipment . . . . . . . . . . . . . . ....
-
b. Your client is considering investing in Bell Limited. The following financial information was provided to you to assist your client with her decision. Operating profit 10% Long Term Loan 2021 ($)...
-
Match the histograms in the next column to the summary statistics given. Standard Deviation Mean Median 53 1.8 53 60 60 11 53 53 III 10 53 53 IV 22 20 - 10 10 32 42 52 62 72 82 48 50 52 54 56 (a)...
-
The syntheses shown here are unlikely to occur as written. What is wrong witheach? 1. Mg 2. * CH3CH2CH3 (a) CH3CH2CH2F C (c) F CH2 Br () CH2 (CH3)2CULI Ether NBS CCl4 " CH3
-
Addition of HBr to a double bond with an ether (?OR) substituent occurs region specifically to give a product in which the ?Br and ?OR are bonded to the same carbon. Draw the two possible carbocation...
-
For the bus arrangement of Figure 9-47, describe the input signal requirements for simultaneously transferring the contents of register C to both of the other registers. Figure 9-47 Only one...
-
Tommy Sharp was an electrical and instrumentation (E&I) designer for Aker Plant Services Group. Sharp began working for Aker as a contract employee in 2003, and then as a full-time employee in 2005....
-
Z Ltd . paid Part I federal tax of $ 4 6 , 1 0 7 on taxable income of $ 4 2 0 , 0 0 0 . Net income for tax purposes consists of $ 3 9 2 , 0 0 0 of active business income and $ 2 8 , 0 0 0 of interest...
-
O'Bannon Publishing Company is an Indiana corporation that is in the business of newspaper publishing and commercial and consumer printing. O'Bannon's printing enterprise is operated under the trade...
-
Urban Glam Cosmetics made purchases of lipstick in the current year as follows: Jan. 1 Beginning inventory 9 5 units @ $ 1 3 . 0 0 = $ 1 , 2 3 5 Mar. 1 4 Purchased 3 5 0 units @ $ 1 4 . 0 0 = 4 , 9 0...
-
Information is critical for decision-making. In the case of the man mentioned in the passage above, the information he has to make his decisions comes from God's Law. In businesses, decisions are...
-
You promised your grandparents to spend the coming weekend with them for the whole day, but in the meantime, your office team outing is planned. What will you do?
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
The azide ion, N 3 , is a symmetrical ion, all of whose contributing resonance structures have formal charges. Draw three important contributing structures for this ion.
-
What would be the product ratio in the chlorination of propane if all the hydrogens were abstracted at equal rates?
-
Determine whether the following objects are chiral or achiral. T 0/
-
If you had the two enantiomers of carvone in unmarked bottles, could you use just your nose and a polarimeter to determine? (a) Whether it is the (+) or (-) enantiomer that smells like spearmint? (b)...
-
(6 pts.) The following gas phase reaction system is conducted in an isothermal constant volume batch reactor. The reactor is charged with pure A with an initial concentration CAO AB ri = k CA B D r2...
-
Based on the NPV and IRR investment criteria, should your company introduce a new candy flavor? 1) Should the cost of a marketing survey be included in your decision? Why or why not? 2) Calculate the...
-
Line spectra for atoms is generated by electronic transitions between quantum energy levels. For the hydrogen atom we established that in the absence of external fields the energy depends only on the...

Study smarter with the SolutionInn App


