Within each set, which compound should show NMR absorptions with the greater chemical shifts? Explain your choices.
Question:
Within each set, which compound should show NMR absorptions with the greater chemical shifts? Explain your choices.
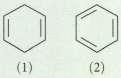
Transcribed Image Text:
(1) (2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (13 reviews)
The aromatic compound has NMR absorptions with gre...View the full answer

Answered By

Mehwish Aziz
What I have learnt in my 8 years experience of tutoring is that you really need to have a friendly relationship with your students so they can come to you with their queries without any hesitation. I am quite hardworking and I have strong work ethics. Since I had never been one of those who always top in the class and always get A* no matter what, I can understand the fear of failure and can relate with my students at so many levels. I had always been one of those who had to work really hard to get decent grades. I am forever grateful to some of the amazing teachers that I have had who made learning one, and owing to whom I was able to get some extraordinary grades and get into one of the most prestigious universities of the country. Inspired by those same teachers, I am to be like one of them - who never gives up on her students and always believe in them!
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Within set, which compound should be more reactive in carbonyl-addition reactions? Explain your choices. H,CCC CH or HCC CH2CH --
-
Within each set, which two structures represent the same compound? A B
-
Which compound in each set should have the larger dipole moment? Explain. Propene or 2-methylpropene
-
For what number does the principal square root exceed eight times the number by the largest amount?
-
What are the disadvantages of using online job sites when you look for your first job?
-
The figure shows a shaft mounted in bearings at A and D and having pulleys at B and C. The forces shown acting on the pulley surfaces represent the belt tensions. The shaft is to be made of ASTM...
-
A study was conducted to measure the effect of 3 different meat tenderizers on the weight loss of steaks having the same initial (precooked) weights. The effects of cooking temperatures and cooking...
-
Consider a two-period model with two firms, A and B. In the first period, they simultaneously choose one of two actions, Enter or do not enter. Entry requires the expenditure of a fixed entry cost of...
-
1. let f(n) = 2 and g(n) = n. Asymptotically, which one grows faster? Prove it. 2. Use induction to prove that the following equation is true for every positive integer n. Show all three steps. 3....
-
The materials manager at Pine Valley Furniture Company maintains a list of suppliers for each of the material items purchased by the company from outside vendors. Table 4-7 shows the essential data...
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
Outline laboratory syntheses of each of the following compounds, starting with benzene and any other reagents. (The references to equations will assist you with nomenclature.) (a) p-dibromobenzene...
-
Graph these cost behaviour patterns over a relevant range of 010,000 units: a. Variable expenses of $8 per unit b. Mixed expenses made up of fixed costs of $20,000 and variable costs of $3 per unit...
-
Chapter 07 Homework D 1. EX.07.07A 2. PR.07.11A ogress: 2/2 items eBook Cash Short and Over Entries Listed below are the weekly cash register tape amounts for service fees and the related cash counts...
-
Klumper Corporation is a diversified manufacturer of industrial goods. The company's activity-based costing system contains the following six activity cost pools and activity rates: Activity Cost...
-
Following is some financial information of Kean Inc. 2022 2021 $49,205 $41,444 Revenues Net income Total assets Shareholder's equity Cash flow from operations Basic earnings per share share Book...
-
L23 7 5 6 8 9 10 11 12 13 5 14 15 16 17 18 19 20 21 22 23 24 25 26 27 B C D ACTG 382 Excel Homework Assignment #1 Winter 2024 A a. Total contract price Instructions b. Company Background Information:...
-
Essentially, I am required to do a self reflective report using Gibbs Reflective cycle Model about my experiences with 'working with the industry' (My module name) . I understand the bullet points...
-
Visit www.pcmag.com/roundup/341251/the-best-robot-vacuums. How do the reviews of iRobot vacuums compare to competitive robot vacuums? Does the information from the review match the value proposition...
-
Find the cross product a x b and verify that it is orthogonal to both a and b. a = (t, 1, 1/t), b = (t 2 , t 2 , 1)
-
The sequence of reactions shown in Fig. P24.52, called the Weerman degradation, can be used to degrade an aldose to another aldose with one less carbon atom. Using glucose as the aldose, explain what...
-
Compound A, known to be a monomethyl ether of D-glucose, can be oxidized to a carboxylic acid B with bromine water. When the calcium salt of B is subjected tothe Ruff degradation, another aldose...
-
The Wohl degradation, shown in Fig. P24.51, can be used to convert an aldose into another aldose with one fewer carbon. Give the structure of the missing compounds as well as the curved-arrow...
-
Janice will need to pay $200 at the end of every month for the next 12 months, except for the payment of the 8th month. What is the present value, assuming a rate of 4%, compounded quarterly?
-
List and explain two management tools in the planning process and two measurable performance indicators. Explain in detail.
-
What does a high PE tell us about the value of the stock price (over or under valued)? What does a low PE tell us about the value of the stock price (over or under valued)? Be specific with your...

Study smarter with the SolutionInn App


