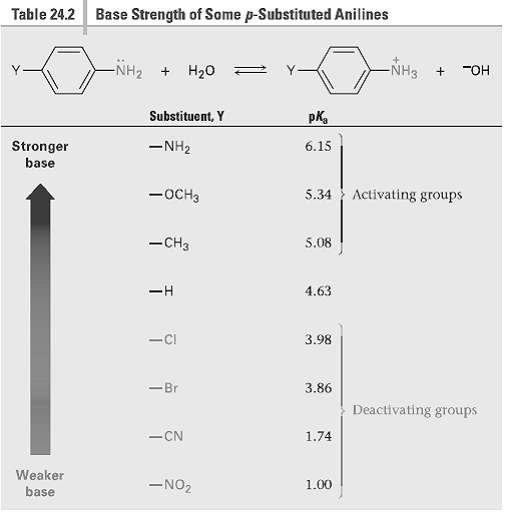
Without looking at Table 24.2, rank the following compounds in order of ascending basicity. (a) p-Nitro aniline,
Question:
Without looking at Table 24.2, rank the following compounds in order of ascending basicity.
(a) p-Nitro aniline, p-aminobenzaldehyde, p-Bromoaniline
(b) p-Chloroanilinc, p-aminoacetophenone, p-methyl aniline
(c) p-(Trifluoromethyl) aniline, p-methyl aniline, p-(fluoro methyl)aniline
Transcribed Image Text:
Table 24.2 Base Strength of Some p-Substituted Anilines -NH2 + H20 NH3 "OH Substituent, Y pKa -NH2 Stronger base 6.15 5.34 Activating groups -OCH3 -CH3 5.08 4.63 3.98 -Ci -Br 3.86 Deactivating groups -CN 1.74 Weaker -NO2 1.00 base
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (19 reviews)
The basicity order of substituted arylamines is the same as their react...View the full answer

Answered By

John Kago
Am a processional practicing accountant with 5 years experience in practice, I also happens to have hands on experience in economic analysis and statistical research for 3 years. am well conversant with Accounting packages, sage, pastel, quick books, hansa world, etc, I have real work experience with Strata, and SPSS
4.70+
31+ Reviews
77+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following compounds in order of decreasing acid strength: CH2OH COOH
-
Rank the following compounds in order of decreasing acidity:
-
Rank the following compounds in order of increasing reactivity (least reactive first) in an SN1 solvolysis reaction in aqueous acetone. Explain your answers. (The structure of tert-cumyl chloride is...
-
2. Cash basis $(3,400)Fantastic Catering completed the following selected transactions during May 2016: May 1 Prepaid rent for three months, $1,800. 5 Received and paid electricity bill, $200. 9...
-
How would you suggest that your department apply these priorities in planning its management training programs?
-
On heating, cis-4-hydroxycyclohexanecarboxylic acid forms a lactone but trans-4-hydroxycyclohexanecarboxylic acid does not. Explain.
-
An auditor expects no errors in an audit population but is concerned about potential monetary overstatement and, therefore, elects to use probability-proportional-to-size sampling. Recorded book...
-
The Runabout Boat Company is considering the purchase of several units of an expensive new machine for its production facilities around the country. Before committing to this purchase, Runabout wants...
-
Iron Sheets Company had the following information for the month of June 2021. Sales Purchases Sales and administrative expenses V Factory overhead V Direct labor Shs. 257,000 92.000 79,000 37,000...
-
Debbie Gibson is considering three investment options for a small inheritance that she has just receivedstocks, bonds, and money market. The return on her investment will depend on the performance of...
-
The benzyl ammonium ion (C6H5CH2NH3) has pKa 9.33, and the propyl ammonium ion has pKa = 10.71. Which is the stronger base, benzyl amine or propylamine? What are the pKbS of benzyl amine and...
-
Calculate the percentages of neutral and protonated forms present in a solution of 0.0010 M pyrimidine at pH = 7.3. The pKa of pyrimidinium ion is 1.3.
-
Find the equivalent resistance between points a and b infigure. 2.4 N 5.1 N 1.8 N 3.5 0 bo- 3.6 N
-
Why is retaliation theory a cause for concern for agencies? What liabilities do companies face?
-
What is the public policy for negligence? If someone does not intentionally cause damage or harm to another, why should they still be held liable under negligence?
-
Samuel has a longstanding interest in cryptocurrency. Can the company raise funds via bitcoins?
-
Discuss the five Rules of Sale of Goods Act with examples. It should not be copy and paste. it should be in own words. Discuss acoording to canadian law.
-
Canada ( Attorney General ) v. Chambre des notaires du Quebec, [2016] 1 SCR 336, 2016 SCC 20 (CanLII) What is the identified legislation related to the case? What is the legal question in the case...
-
In a photoelectric effect experiment, the intensity of the light is increased while the frequency, which is above the threshold frequency, is held constant. As a result, A. There are more electrons....
-
Below is a sample of the data in the file NFLAttendance which contains the 32 teams in the National Football League, their conference affiliation, their division, and their average home attendance....
-
When N 2 O 5 (g) is heated, it dissociates into N 2 O 3 (g) and O 2 (g) according to the reaction: The N 2 O 3 (g) dissociates to give N 2 O(g) and O 2 (g) according the reaction: When 4.00 mol of N...
-
Polyethylene glycol, or Carbowax [1 - O - CH2 - CH2 -)n] is widely used as a binder, thickening agent, and packaging additive for foods. (a) What type of polymer is polyethylene glycol? (We have not...
-
Ring-opening metathesis polymerization (ROMP, see Section 8-17) is a promising new technique for polymerizing cyclic olefins. In its simplest form, the reaction involves a cycloalkene (preferably...
-
Polyoxymethylene (polyformaldehyde) is the tough, self-lubricating Delrin plastic used in gear wheels. (a) Give the structure of polyformaldehyde. (b) Formaldehyde is polymerized using an acidic...
-
Harwood Company uses a job-order costing system that applies overhead cost to jobs on the basis of machine-hours. The company's predetermined overhead rate of $2.60 per machine-hour was based on a...
-
Dahlia Corporation has a current accounts receivable balance of $447,016. Credit sales for the year just ended were $4,950,605. a. What is the receivables turnover? Note: Do not round Intermediate...
-
Crenshaw Inc. is considering a change to activity-based product costing. The company produces two products, cell phones and tablet PCs, in a single production department. The production department is...

Study smarter with the SolutionInn App


