Would you expect the following carboxylic acids to be more acidic or less acidic than benzoic acid?
Question:
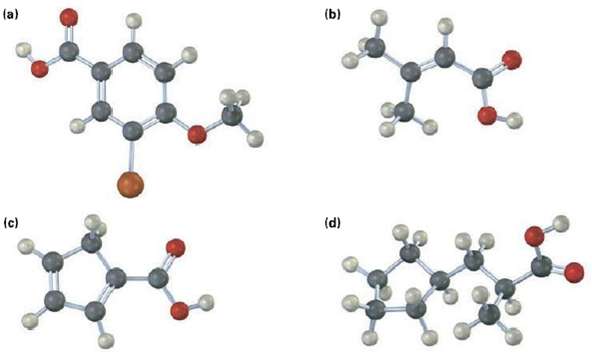
Would you expect the following carboxylic acids to be more acidic or less acidic than benzoic acid? Explain. (Reddish brown = Br)
Transcribed Image Text:
(a) (b) (d) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Br COH b COH CH32N a pBromobenzoic acid is more acidic th...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Would you expect the citric acid cycle to be more or less active when a cell has a high ATP/ADP ratio and a high NADH/NAD+ ratio? Give the reason for your answer.
-
Would you expect the following compound to be aromatic? Explain your answer.
-
Would you expect the substituent? to more closely resemble in its effect on rate and regioselectivity in electrophilic aromatic substitution? Why? N(CH N(CH3)2 or NO2
-
Read the Poem Little Birds Flying and answer the following questions: What it is notifying? To whom it is notifying? How is the work two dimensional? What does it take to realize the project? ...
-
1. What unique challenges did Rande and Jonas face when entering into this partnership? 2. What impact did their friendship seem to have on this partnership? 3. What partner duties did Jonas fail to...
-
The table gives enrollments at North Shore High School. a. Find the equation of the regression line. Round the slope and y-intercept to the nearest hundredth. b. What is the slope of the linear...
-
What are examples of Human Systems Environment threats and their sources?
-
Sweet Catering completed the following selected transactions during May 2014: May 1 Prepaid rent for three months, $ 1,500. 5 Received and paid electricity bill, $ 400. 9 Received cash for meals...
-
Fill out the daily spending diary for a 51-year-old woman with two kids, spending $1600 on rent, not eating out frequently, purchasing groceries, and having an electric car to minimize gas expenses....
-
What is the reaction of standard TCP in case of packet loss? In what situation does this reaction make sense and why is it quite often problematic in the case of wireless networks and mobility?
-
Give IUPAC name for the following carboxylic acids (reddish brown = Br). (a) (b) (d) (c)
-
The following carboxylic acid can?t be prepared from an alkyl halide by either the nitrile hydrolysis route or the Grignard carboxylation route. Explain.
-
Use the formula or a calculator application to find the monthly payment on a home mortgage of $645,730 at 4.862% interest for 20 years?
-
Many of you have worked for or experienced various types of leaders. Identify one major quality you believe makes a good leader and explain how this quality interrelates to one values? Please explain...
-
Accountants, Ethics, and the Financial Statements ( 2 5 Marks ) For some time, Hernadez Pty Co . a beverage manufacturer has been selling to the west coast region, however recently their market share...
-
Rose Apothecary Inc. Issued a 3 - year $ 1 0 0 , 0 0 0 bond with an 8 % stated rate sold to yield 1 0 % on January 1 , 2 0 2 4 . Interest is payable annually on December 3 1 st . Bond Issue Costs are...
-
Ralph contributes land with a basis and value of $ 1 2 0 , 0 0 0 in exchange for a 3 0 % share of the profits and losses in Orange LLC . Orange is a calendar year LLC . Orange generates $ 3 0 0 , 0 0...
-
Tax on purchases during the year Interest from a savings account Interest from City of Boston Bonds Dividend from 3 M stock $ 1 , 4 0 0 5 0 0 9 , 0 0 0 2 , 6 0 0 1 , 0 0 0 6 , 2 0 0 8 0 0 7 0 0 4 0 0...
-
In Example 6.1, we introduced the Hermitian number and phase operators \(\hat{N}\) and \(\hat{\Theta}\) constructed from the raising and lowering operators, \(\hat{a}^{\dagger}\) and \(\hat{a}\). In...
-
What can you do to reduce hunger where you live? To reduce hunger globally?
-
Polyacetylene is an addition polymer with the structure shown here. Draw the structure of the monomer. H [] C. C H
-
(a) What are the structures of L-(+)-threose and L-(+)-erythrose? (b) What aldotriose would you use to prepare them in a Kiliani-Fischer synthesis?
-
(a) Outline a Kiliani-Fischer synthesis of epimeric aldopentoses starting with D-(-)- erythrose (use Fischer projections). (b) The two epimeric aldopentoses that one obtains are D-(-)-arabinose and...
-
Subjecting D-(-)-threose to a Kiliani-Fischer synthesis yields two other epimeric aldopentoses, D-(+)-xylose and D-(-)-lyxose. D-(+)-Xylose can be oxidized (with nitric acid) to an optically inactive...
-
Total Labor- Square Feet of Department Hours Space Occupied Number of Employees Machine- Hours Direct Labor- Hours Personnel 16,000 12,400 22 Custodial Services 8,200 3,100 49 Maintenance 14,300...
-
6. Given the Venn Diagram, find (AUB)NC A B a W b Y N X C C
-
Prepare one-page description of the company and a one-page description of the project. In the project description, please list all the key stakeholders who will attend the project kickoff meeting....

Study smarter with the SolutionInn App


