Would you expect the following reaction to proceed in a conrotatory or disrotatory manner? Show the stereochemistry
Question:
Would you expect the following reaction to proceed in a conrotatory or disrotatory manner? Show the stereochemistry of the Cyclobutene product, and explain youranswer.
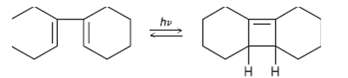
Transcribed Image Text:
hv
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
A photochemical electrocyclic reaction involving two ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assume that SCR" is a convex set and that g: S R. Show that the set g(x) 0 is convex if g is convex. What about the opposite implication?
-
Would you expect the 2-octanol formed by SN2 hydrolysis of (+) - 2-bromooctane to be optically active? If so, what will be its absolute configuration and sign of rotation? What about the 2-octanol...
-
Would you expect the substituent N (CH3)3 to more closely resemble N (CH3)2 or NO2 in its effect on rate and regioselectivity in electrophilic aromatic substitution? Why?
-
Why do you suppose P&Gs marketing research includes face-to-face interviews for the situation described in this case? Would other forms of marketing research also be useful in this situation? Explain...
-
Critics of activist investors argue that they force firms to focus on short-term considerations versus long-term performance. As such, they believe that shareholder rights to remove board members, to...
-
Use the data in MURDER.RAW for this exercise. The variable mrdrte is the murder rate, that is, the number of murders per 100,000 people. The variable exec is the total number of prisoners executed...
-
The van der Waals equation of state is given by (a) \(\left(P+\frac{a}{V} ight)(V-b)=R T\) (b) \(\left(P+\frac{a}{V^{2}} ight)(V-b)=R T\) (c) \(\left(P+\frac{a}{V^{2}} ight)(V+b)=R T\) (d) None of...
-
The trial balance of Mr. Eko Fashion Center contained the following accounts at November 30, the end of the company?s fiscal year. Adjustment data:1. Supplies on hand totaled $2,000.2. Depreciation...
-
8% Intermediate Accounting Co. Shareholders' equity 31-Dec-19 Preferred shares, no par value, cumulative and participating 1,000 shares issued & outstanding Common shares, no par value, 100,000...
-
A home-team baseball victory produces benefits for residents (in terms of hometown pride) that is both nonrival and nonexcludable. The graph on the right depicts the marginal benefits that Beatrice,...
-
The following thermal isomerization occurs tinder relatively mild conditions. Identify the pericyclic reactions involved, and show how the rearrangementoccurs. CH C- CeH5. C&H5 "CH CD CoH5 CgH5 CEH5...
-
Heating (1Z, 3Z, 5Z)-1, 3, 5-cyclorionatriene to 100?C causes Cyclization and formation of a bicyclic product. Is (he reaction conrotatory or disrotatory? What is the stereo chemical relationship of...
-
The O'Hannigan Company of Edmonton made the following transactions relating to property, plant, and equipment during 2012. Use the company's General Journal to record the following transactions: 2012...
-
In the field of ethics, what is a conflict of interest and what are some things that can to done to minimize conflicts of interest? Of all the issues in the article, what did you find to be the most...
-
Will your company warranty your products? Why or why not? What, if any, express warranties will your company offer? To what, if any, implied warranties will your company be subject? If you are not...
-
The grand management paradox that systems thinkers face is a result of the tension that exists between the forces of static equilibrium or the resistance to change and dynamic equilibrium or being...
-
Project: Drafting a Bill You are concerned about a matter that has federal implications and is tremendously important in your city. You would like to meet with your US Representative or Senator to...
-
Many large companies are now taking their Ethical futures very seriously. However, I also believe that some, whilst projecting the image of having good Ethical positions, view this more from a...
-
If you eat a pound of pasta, will you gain a pound of weight? Why not?
-
Which of the companies has the lowest accounts receivable turnover in the year 20X2? a. Company A. b. Company B. c. Company C. d. CompanyD. 20X1 20X2 Credit Sales Average Receivables Balance $1.0...
-
Congress and the SEC have attempted to enhance market competition among U.S. trading venues without intensifying the harmful effects of market fragmentation. What is market fragmentation, and what...
-
There is an overall 29-fold difference in reactivity of 1-chlorohexane, 2-chlorohexane, and 3-chlorohexane toward potassium iodide in acetone. (a) Which one is the most reactive? Why? (b) Two of the...
-
In each of the following indicate which reaction will occur faster. Explain your reasoning. (a) CH3CH2CH2CH2Br or CH3CH2CH2CH2I with sodium cyanide in dimethyl sulfoxide (b) 1-Chloro-2-methylbutane...
-
Under conditions of photochemical chlorination, (CH3)3CCH2C(CH3)3 gave a mixture of two monochlorides in a 4:1 ratio. The structures of these two products were assigned on the basis of their SN1...
-
MM Corp. has 50,000 shares outstanding with share price of $18. It has debt with market value of $300,000. The equity beta is 1.2 and debt beta is 0.1. The risk-free rate is 2% and the market risk...
-
The market is expected to return 15 percent next year and the risk-free rate is 7 percent. What is the expected rate of return on a stock with a beta of 1.3? The covariance of the market's returns...
-
A stock's current price is 145.05. A put option with an exercise price of 120 and maturity of 3 months is currently priced at $ 28.83. What is the option's time value?

Study smarter with the SolutionInn App


