Write the equations for the remaining passages of the p-oxidation pathway following those shown infigure. CHCH2CH2CHH-CH2 CH2CH2CH2CH2
Question:
Write the equations for the remaining passages of the p-oxidation pathway following those shown infigure.
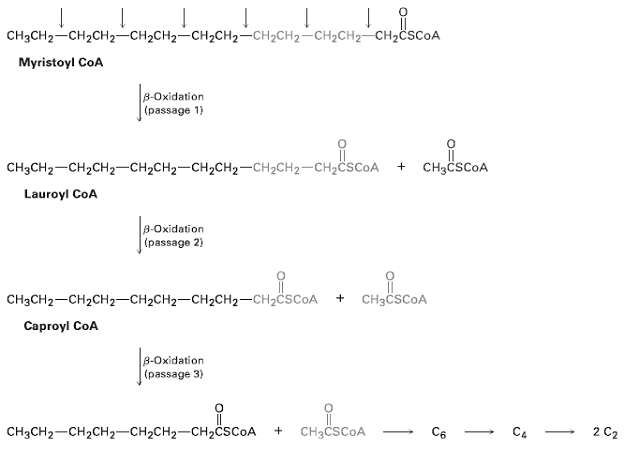
Transcribed Image Text:
CHяCH2—CH2CH—сH-CH2— CH2CH2—CH2CH2— CH2CH2 CH-CSCOA Myristoyl CoA 8-Oxidation (passage 1) CH3CH2-CH2CH2-CH2CH2-CH2CH2--CH2CH2-CH2CSCOA + CH3CSCOA Lauroyl CoA B-Oxidation (passage 2) CH3CH2-CH2CH2-CH2CH2-CH2CHz-CH2CSCOA + CH3CSCOA Caproyl CoA 8-Oxidation (passage 3) CH3CH2-CH2CH2-CH2CH2-CH2CSCOA CH3CSCOA C4 - 2 C2 Св
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
CH3CHCHCHCHCHCHCSCOA Caprylyl CO...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Write the equations for the 95% confidence limits, and solve them numerically if you have a computer. 14 mutations are counted in one million base pairs.
-
Write the equations for the approximate 95% confidence limits using the method of support, and solve them numerically if you have a computer. 14 mutations are counted in one million base pairs.
-
Write the equations for the carry-lookahead logic for a 64-bit adder using the new notation from Exercise B.26 and using 16-bit adders as building blocks. Include a drawing similar to Figure B.6.3 in...
-
In programming challenge 8 of chapter 3, you created an application that converts celsius temperatures to fahrenheit. recall that the formula for performing this conversion isf = 1.8 * c + 32in the...
-
Identify the form of payment, form of acquisition, acquisition vehicle, and post-closing organization? Speculate why each may have been used.
-
Use the data in LAWSCH85.RAW for this exercise. (i) Using the same model as Problem 3.4, state and test the null hypothesis that the rank of law schools has no ceteris paribus effect on median...
-
The activity of a component depends on (a) Temperature only (c) Temperature, pressure and composition (b) Temperature and pressure only (d) None of these.
-
A process that is considered to be in control measures an ingredient in ounces. Below are the last 10 samples (Each of size n = 5) taken. The population standard deviation is 1.36. (a) What is the...
-
How do environmental factors, including industry volatility, technological disruption, and geopolitical instability, intersect with organizational commitment, shaping employee attitudes and behaviors...
-
Shauna Coleman is single. She is employed as an architectural designer for Streamline Design (SD). Shauna wanted to determine her taxable income. She correctly calculated her AGI. However, she wasnt...
-
One of the steps in fat metabolism is the reaction of glycerol (1, 2, 3-propanetriol) with ATP to yield glycerol 1-phosphate. Write the reaction, and draw the structure of glycerol 1-phosphate.
-
How many molecules of acetyl CoA are produced by catabolism of the following fatty acids, and how many passages of the a-oxidation pathway arc needed? (a) Palmitic acid, CH 3 (CH 2 ) 14 CO 2 H (b)...
-
Does the molecular model shown represent (-)-2-butanol or (-)-2-butanol?
-
How do alterations in epigenetic modifications, including DNA methylation, histone acetylation, and chromatin remodeling, contribute to the pathogenesis of complex diseases such as cancer,...
-
Regulation and deregulation is a big deal in modern politics. Whether its "drain the swamp," or Boeing's freedom to do what it wants to do, or the Biden emphasis on controlling Wall Street and...
-
Which of the following accurately describes something that occurs with the 2- component regulatory system? O Binding of a signal molecule to the sensor causes a phosphate group to fall off the...
-
How does an organization incorporate strategic goals and initiatives into its budget? how can true strategic initiatives be identified ? What evidence can show that their implementation is budgeted?
-
Every player has a 52-card deck and draws a card at random from it. Simultaneously the players decide to bet or pass. If both bet, they show their card and the player with the highest value wins...
-
A \(0.50-\mathrm{kg}\) wood block initially traveling at \(1.0 \mathrm{~m} / \mathrm{s}\) slides \(0.50 \mathrm{~m}\) on a horizontal floor before coming to rest. What is the average rate at which...
-
General Electric Capital, a division of General Electric, uses long-term debt extensively. In a recent year, GE Capital issued $11 billion in long-term debt to investors, then within days filed legal...
-
Redo Problem 13.1 using Aspen Plus. Problem 13.1 Isopropyl alcohol is to be dehydrogenated in the gas phase to form propionaldehyde according to the reaction Compute the equilibrium fraction of...
-
Based on what you know about disubstituted cyclohexanes, which of the following two stereoisomeric 1,3,5-trimethylcyclohexanes would you expect to be more stable? CH3 CH3 H CH3...
-
Write structural formulas, and give the functional class and substitutive names of all the isomeric alkyl chlorides that have the molecular formula C4H9Cl.
-
Is the equilibrium constant for proton transfer from hydrogen chloride to tert-butyl alcohol greater than 1 or less than 1?
-
What are the challenges and strategies for implementing TPM in highly regulated industries, such as pharmaceuticals or aerospace? How can TPM contribute to compliance and quality assurance in these...
-
In multi-site manufacturing operations, what role does TPM play in standardizing maintenance practices and ensuring consistency across different plants or facilities? Discuss the challenges and...
-
Identify and evaluate the components of a comprehensive total rewards and motivation system? Illustrate with specific examples.

Study smarter with the SolutionInn App


