A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an
Question:
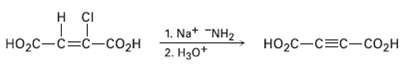
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that (Z)-2-chloro-2-butenedioic acid reacts 50 times as fast as the corresponding E isomer. What conclusion can you draw about the stereochemistry of eliminations in vinylic halides? How does this result compare with eliminations of alkylhalides?
Transcribed Image Text:
H CI |НO2с — С%3с—содн 1. Na* "NH2 2. Hзо* но-с—с3с-соон
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (12 reviews)
COH Z2Chloro2butene 14dioic acid HOC H HOC E2Chloro2butene 14dioic acid Hydrogen a...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many different license plate numbers can be made by using one letter followed by ve digits selected from the digits 0 through 9?
-
As the drawing illustrates, a siren can be made by blowing a jet of air through 20 equally spaced holes in a rotating disk. The time it takes for successive holes to move past the air jet is the...
-
A classic homemade ice cream can be made by freezing the ice cream mixture using a solution of 250 g of NaCl dissolved in 1.25 kg of ice water. What is the temperature of this ice water? Assume an...
-
Zebra Corporation has always been an S corporation and is 100% owned by Paul. Paul has a basis of $40,000 in his Zebra stock at the beginning of the year. During the year, Zebra has an ordinary loss...
-
Johnson and Johnson has been highly successful using a decentralized structure. What are some of the risks associated with decentralization? How do you maintain a decentralized structure but provide...
-
The account balances of Islewood Ltd. and Richmond Ltd. at January 1, 2013, were as follows: The fair values of Richmonds assets at January 1, 2013, were: Land .............240,000...
-
You are short 20 gasoline futures contracts, established at an initial settle price of .545 (see Figure 16.1 for contract specifications). Your initial margin to establish the position is $1,200 per...
-
The internal control procedures in Phillips Company make the following provisions. Identify the principles of internal control that are being followed in each case. (a) Employees who have physical...
-
On June 1, 2018, Bridgeport Company and Indigo Company merged to form Sweet Inc. A total of 764,000 shares were issued to complete the merger. The new corporation reports on a calendar-year basis. On...
-
Based on the following transactions, calculate the revenues, expenses, net income that would be reported (a) On the cash basis and (b) On the accrual basis i. Purchased inventory with a cost of...
-
Reaction of iodoethane with CN yields a small amount of isonitrile, CH3CH2N C, along with the nitrile CH3CH2C N as the major product. Write electron-dot structures for both products, assign formal...
-
(S)-2-Butanol slowly racemizes on standing in dilute sulfuric acid.Explain. CH3CH2CHCH3 2-Butanol
-
Find the equation of the plane with perpendicular n = (1, 1, 1) that passes through the point with position vector (2, 3, 3). Show that the line with equation r = (1, 1, 2) + t(2, 0, 2) lies in this...
-
See the following information. Sales Account Payable Long-term Debt Account Receivable Balance Sheet information Cash $300,000 $5,000000 $600,000 $1,300,000 $700,000 Inventory $1,500,000 Long-term...
-
Carrefour is a large multinational supermarket brand that is present in many different markets (countries) in Europe and Asia. Carrefour has a private (store) brand. What are the private branding...
-
- Given Q80 3P+0.01Y, where Q is quantity demanded, P is price, and Y is income, and given P = 25 and Y = 3000, find the 1. Price elasticity of demand. 2. Income elasticity of demand.
-
At the end of 1993, the exchange rate between Mexico and the United States was 3.1 pesos to the dollar. By the end of 1994, the exchange rate was 5 pesos to the dollar, and at the end of 1995, it was...
-
Mr. Jackie Chan and Mr. James Bond are partners who shared profit and losses on an arbitrary ratio of 3:2. Mr. Jet Lee was admitted into the partnership by buying 1/4 and 1/2 of the respective old...
-
The following TI-84 Plus display presents the results of a test of the null hypothesis H0 : 1 = 0. a. What is the alternate hypothesis? b. What is the value of the test statistic? c. How many degrees...
-
The relationship described in question 7 does not always appear to hold. What factors, besides the number of firms in the market, might affect margins?
-
What is the effect of a temperature change on a chemical reaction initially at equilibrium? How does the effect differ for an exothermic reaction compared to an endothermic one?
-
A student intends to carry out the following synthesis: CH2 Ph (1) base (2) PhCH, Br
-
Some (but not all) of the following keto esters can be formed by Dieckmann condensations. Determine which ones are possible, and draw the starting diesters. (a) (b) (c) (d) C-OCH,CH
-
Propose a mechanism for the crossed Claisen condensation between ethyl acetate and ethyl benzoate.
-
Consider how Star Valley, a popular ski resort, could use capital budgeting to decide whether the $8 million Blizzard Park Lodge expansion would be a good investment. Assur conce assur Click the icon...
-
Selected T-accounts of Moore Company are given below for the just completed year: Raw Materials Debit Credit Balance 1/1 28,000 Credits Debits 146,000 Balance 12/31 38,000 Manufacturing Overhead...
-
Your company has total receivables outstanding at 31 December 2020 of $20,000. You think that about 2% of these balances will not be collected and wish to make an appropriate allowance. You have not...

Study smarter with the SolutionInn App


