A compound A (C6H) undergoes catalytic hydrogenation over Lindlar catalyst to give a compound B, which in
Question:
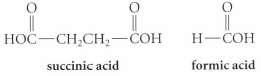
A compound A (C6H) undergoes catalytic hydrogenation over Lindlar catalyst to give a compound B, which in turn undergoes ozonolysis followed by workup with aqueous H2O2 to yield succinic acid and two equivalents of formic acid. In the absence of a catalyst poison, hydrogenation of A gives hexane. Propose a structure for compound A.
Transcribed Image Text:
HOC-CH,CH2-СОН Н-СОН succinic acid formic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (18 reviews)
The ozonolysis results define compound B as 1 5hexadi...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
An optically active monoterpene (compound A) with molecular formula undergoes catalytic hydrogenation to form an optically inactive compound with molecular formula C10H20O (compound B). When compound...
-
Compound A undergoes catalytic hydrogenation much faster than does compound B. Why? Making molecular models will help.
-
Compound A undergoes catalytic hydrogenation much faster than does compound B. Why? Making molecular models will help. .. .. B.
-
Able Control Company, which manufactures electrical switches, uses a standard cost system and carries all inventory at standard cost. The standard factory overhead cost per switch is based on DLHs. *...
-
Which one of the following statements is not an advantage of decentralization? a. Segment managers usually have more information regarding matters within their segments than do managers at higher...
-
On December 31, 2016, Clarke, Inc. borrowed \(\$ 900,000\) on a seven percent, 10 -year mortgage note payable. The note is to be repaid in equal annual installments of \(\$ 128,140\) (payable on...
-
The Lorton Company acquired land containing coal. Lorton will restore the land to a condition suitable for recreational use after it has extracted the coal. Geological surveys estimate that the...
-
You would like to accumulate $ 1 5 , 5 0 0 by the end of three years from now to buy a used car. You have $ 2 , 5 0 0 now and would like to save equal annual end - of - year deposits to pay for the...
-
Flowmaster Forge Inc. is a designer and manufacturer of industrial air-handling equipment that is a wholly owned subsidiary of Howden Industrial Inc. Howden is interested in selling Flowmaster to an...
-
Propose mechanisms for each of the following known transformations: use the curved-arrow notation where possible. NaOD D2O (large excess) THF Ph--C-D Ph-CEC-HH
-
Complete the reactions given in Fig. P14.45 using knowledge or intuition developed from this or previous chapters. (a) (b) CH CH MgBr DO CH,CH CH CH2 CH 0 CH CH2-O S-O- CH2CH3 diethvl sulfate
-
A coil develops 800 cal/s when 20 V is supplied across its ends. Compute its resistance.
-
A company currently has $265,000 of equity and is planning a $115,000 expansion. The company currently earns $79,500 in net income, and the expansion will yield $50,000 in additional income before...
-
Packaging Solutions Corporation manufactures and sells a wide variety of packaging products. Performance reports are prepared monthly for each department. The planning budget and flexible budget for...
-
Write a program that reads maximum 100 integers entered by user ranging from 1 to 9 and displays the following report. Enter ten numbers: 1 2 3 2 1 6 5 5 72894 (Enter) The number of distinct number...
-
(a) Show that the three vectors V = (0, 3, 1, -1), V2 = (6, 0, 5, 1), and v3 = (4, -7, 1, 3) form a linearly dependent set in R4. (b) Express each vector in part (a) as a linear combination of the...
-
Find the indicated partial derivatives. f(x, y) = 8xy + x7y'; fxxy fyyy fxxy(x, y) fyyy(x, y) = Need Help? Read It Watch It
-
Perform the indicated operations. Is a 2 < a 1 for any negative value of a? Explain.
-
A manufacturer can sell product 1 at a profit of $20 per unit and product 2 at a profit of $40 per unit. Three units of raw material are needed to manufacture one unit of product 1, and six units of...
-
How would you prepare the following compounds starting with an appropriate carboxylic acid and any other reagents needed? (Reddish brown =Br.) (a) (b)
-
The following structure represents a tetrahedral alkoxide-ion intermediate formed by addition of a nucleophile to a carboxylic acid derivative. Identify the nucleophile, the leaving group, the...
-
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which of the two do you think is more reactive in nucleophilic acyl substitution reactions?...
-
ty- Calculating the WACC Cost of new common stock: 15.71 % b. Now calculate the cost of common equity from retained earnings, using the CAPM method. 13.02 % c. What is the cost of new common stock...
-
Rex acquired 80% of the voting common stock of Sam Enterprises on January 1, 2025. During 2025, Sam Enterprises had revenues of $2,700,000 and expenses of $2,100,000. The amortization of fair value...
-
Cantoon Co . is considering the acquisition of a unit from the French government. Its initial outlay would be $ 3 . 5 million. It will reinvest all the earnings in the unit. It expects that at the...

Study smarter with the SolutionInn App


