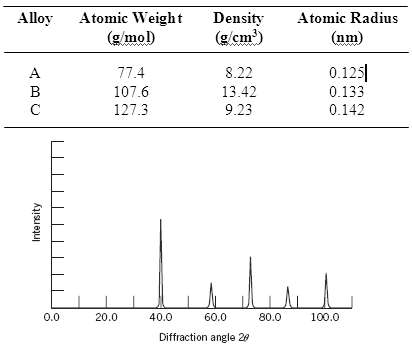
Below are listed the atomic weight, density, and atomic radius for three hypothetical alloys. For each determine
Question:
Below are listed the atomic weight, density, and atomic radius for three hypothetical alloys. For each determine whether its crystal structure is FCC, BCC, or simple cubic and then justify your determination. A simple cubic unit cell is shown in Figure 3.24.
Transcribed Image Text:
Alloy Atomic Weight Density (g/cm') Atomic Radius (g/mol) (nm) 0.125| 8.22 77.4 107.6 13.42 0.133 127.3 9.23 0.142 0.0 20.0 40.0 60.0 80.0 100.0 Diffraction angle 20 Intersity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 65% (20 reviews)
For each of these three alloys we need by trial and error to ca...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
The atomic weight, density, and atomic radius for three hypothetical alloys are listed in the following table. For each, determine whether its crystal structure is FCC, BCC, or simple cubic and then...
-
Damocles Company manufactures fine swords. Below are listed the net changes in the companys balance sheet accounts for the past year: The following additional information is available about last...
-
If the pattern shown in Figure 3 is continued indefinitely what fraction of the original square will eventually be painted? Figure 3
-
Rocket Science A rocket has total mass Mi = 360 kg, including 330 kg of fuel and oxidizer. In interstellar space it starts from rest, turns on its engine at time t = 0, and puts out exhaust with...
-
Why is a soft, underinflated football at sea level much firmer when it is taken to a high elevation in the mountains?
-
Draw a pie graph for the data in Exercise 3 showing the sales of each item, and analyze the results.
-
How does management determine its critical (key) success factors?
-
Official Reserve Rare Coins (ORRC) was formed on January 1, 2016. Additional data for the year follow: a. On January 1, 2016, ORRC issued no par common stock for $450,000. b. Early in January, ORRC...
-
Crane Ltd. purchased a delivery truck on January 1, 2021, at a cost of $83,920. The truck is expected to have a residual value of $8,070 at the end of its 4-year useful life. Crane has a December 31...
-
Assume that you are the CEO of a major producer of potato chips. You have four plants and discover that one of the plants is more productive than the other three; specifically, the midnight to 8 a.m....
-
Rhodium has an atomic radius of 0.1345 nm and a density of 12.41 g/cm 3 . Determine whether it has an FCC or BCC crystal structure.
-
The unit cell for tin has tetragonal symmetry, with a and b lattice parameters of 0.583 and 0.318 nm, respectively. If its density, atomic weight, and atomic radius are 7.30 g/cm 3 , 118.69 g/mol,...
-
Kalinas Doll Company produces handmade dolls. The standard amount of time spent on each doll is 2.0 hours. The standard cost of labor is $10 per hour. The company planned to make 8,000 dolls during...
-
Should Co-Op Bank phase out the unprofitable Independent Financial Advise /Insurance, and Pathfinder products?
-
Sam is considering a new business venture operating a carrier business. He can buy a van for $60,000 including taxes. He expects it will generate annual revenue of $25,000 for the next 5 years. The...
-
Creepers, Inc., manufactures stuffed spiders and mummies. During September the following information was gathered: Spiders Mummies Units sold 6900 3100 Budgeted sales (units) 7500 2500 Contribution...
-
The company is looking to invest $800,000 for two addition lines for production.Our current ROI targets are at 15%.These two lines would then produce annual profits of $65,000 for each line.Do you...
-
1 . How would you calculate the loss of coastal land due to natural causes into your accounting books? 2 . how would this affect the property value of the house & land? 3 . is there any law or...
-
The times from a high school bell schedule are listed. In Exercises 1520, determine the level of measurement of the data set. Explain your reasoning. 8:00 A.M. 10:44 A.M. 8:52 A.M. 8:56 A.M. 9:48...
-
How can a promoter avoid personal liability for pre-incorporation contracts?
-
Explain why L 100 R 100 for (x) = x 2 on [3, 7].
-
For each of the following stacking sequences found in FCC metals, cite the type of planar defect that exists: (a) . . . A B C A B C B A C B A . . . (b) . . . A B C A B C B C A B C . . . Now, copy the...
-
(a)Using the intercept method, determine the average grain size, in millimeters, of the specimen whose microstructure is shown in Figure 4.14(b); use at least seven straight-line segments. (b)...
-
(a) Employing the intercept technique, determine the average grain size for the steel specimen whose microstructure is shown in Figure 9.25(a); use at least seven straight-line segments. (b) Estimate...
-
Why do investments in long-term companies tend to result in more capital for investors? Question 34 Select one: a. higher tax rates b. lower tax rates c. long-term companies have less overhead costs...
-
Using the guidelines covered in Lesson 3, identify which sentence of each pair is correct. The company president announced that Beijing was the site for the manufacturing expansion. The company...
-
Which of the following statements is true? Question 46Select one: a. Rent payments for a mobile home, made to a community development corporation, can be subtracted from Oregon income b. Amounts...

Study smarter with the SolutionInn App


