How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
Question:
How would you carry out the following transformations?
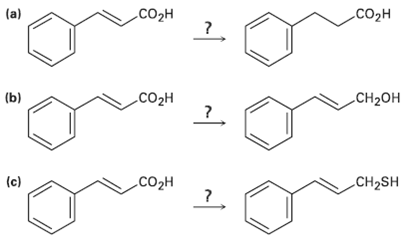
Transcribed Image Text:
Coорн .CO2H (a) (b) Coон CH2он Cоон (c) CH2SH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
In these compounds you want to reduce some but not all of the functional group...View the full answer

Answered By

PU Student
cost accounting
financial accounting
auditing
internal control
business analyst
tax
i have 3 years experience in field of management & auditing in different multinational firms. i also have 16 months experience as an accountant in different international firms. secondary school certification.
higher secondary school certification.
bachelors in mathematics.
cost & management accountant
4.80+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
How would you carry out the following transformations? "Co CH2 C Lc
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
On January 1, 2014, Iron Mountain Ski Corporation purchased a new snow-grooming machine for $50,000. The machine is estimated to have a 10-year life with a $2,000 salvage value. What journal entry...
-
Describe the market requirements and product characteristics in global competition.
-
NorthAm Trucking is a long-haul trucking company serving customers all across the continental United States and parts of Canada and Mexico. At present, all billing activities from preparation to...
-
Who was the auditor for HealthSouth during their fraudulent period? a. Ernst \& Young b. Arthur Andersen c. \(\mathrm{PwC}\) d. Deloitte \& Touche e. KPMG
-
Menge Company has accounts receivable of $93,100 at March 31. An analysis of the accounts shows the following information. Month of Sale Balance, March 31 March .......... $60,000 February ............
-
Stephen, age 40, is single and is not an active participant in a qualified retirement plan. His modified AGI is $57,000 in 2020. The maximum amount that Stephen can deduct for a contribution to a...
-
Many drivers of cars that can run on regular gas actually buy premium in the belief that they will get better gas mileage. To test that belief, we use 10 cars from a company fleet in which all the...
-
What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities. (b) CH |(a) (c) H CHCH2CH2H2H2 C CHCH-CH
-
What carbonyl compounds might you start with to prepare the following compounds by Grignard reaction? List all possibilities. (a) 2-Methyl-2-propanol (b) 1-Ethylcyclohexanol (c) 3-Phenyl-3-pentanol...
-
Practice calculating the sample size and the number of expected errors in attributes sampling by using the tables in Exhibit and the following combinations ofinputs: Tolerable Rate of x Sample Size...
-
Which statement is true when an account is disconnected from the bank feed in QuickBooks Online? Transactions that haven't been categorized yet will be excluded QuickBooks Online will pause the bank...
-
Sheffield corporations December 3 1 , 2 0 2 7 balance sheet showed the following 9 % preferred stock, $ 2 0 par value, cumulative, 1 1 , 0 0 0 shares authorized 9 5 0 0 shares issued $ 1 9 0 , 0 0 0...
-
Please answer and explain clearly the following. Please use at least two (2) strong/robust reference to sustain your answers. At the bottom of your paper, please include the references used, applying...
-
Research the forensic psychology field and find 10 Scholarly references on forensic psychology (including your course Text & psychology.org for forensic info) please list articles or sources below ?
-
Differentiate between the broad and narrow definitions of forensic psychology. Why is it important to distinguish between these two definitions? Choose two of the forensic psychology subspecialties...
-
The most common annual report required by the SEC is Form 10-K. Form 10 , another SEC report, is often used to register under the Securities Exchange Act of 1934. Item 2 of both Form...
-
According during to the IRS, individuals filing federal income tax returns prior to March 31 received an average refund of $1,088 in 2018. Consider the population of "last-minute" filers who mail...
-
We have learned that the enthalpy of vaporization of a liquid is generally a function of temperature. If we wish to take this temperature variation into account, we cannot use the ClausiusClapeyron...
-
Write structural formulas for all of the stereoisomers of 1, 3-dimethylcyclopentane. Label pairs of enantiomers and meso compounds if they exist.
-
Write formulas for all of the isomers of each of the following. Designate pairs of enantiomers and achiral compounds where they exist. (a) 1-Bromo-2-chlorocyclohexane (b) 1-Bromo-3-chlorocyclohexane...
-
Write bond-line three-dimensional formulas for the starting compound, the product, and all of the intermediates in a synthesis similar to the one just given that relates the configuration of...
-
A taxpayer placed a vehicle into service in TY2022 and claimed bonus depreciation of $19,200 on the vehicle. What is the maximum amount of depreciation that can be claimed on the vehicle in TY2023?...
-
If the Inventories account on a balance sheet is $4,238, the Total Current Assets of the same balance sheet is $10,398, and the Total Assets is $18,892, what percent would show for the Inventories...
-
The budgeted income statement does not contain data from the Blank______. Multiple choice question. cash budget ending balance sheet sales budget beginning balance sheet

Study smarter with the SolutionInn App


