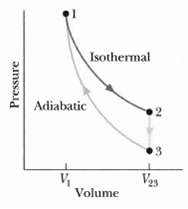
In Figure where V23 = 3.00V1, n moles of a diatomic ideal gas are taken through the
Question:
In Figure where V23 = 3.00V1, n moles of a diatomic ideal gas are taken through the cycle with the molecules rotating but not oscillating. What are?
(a) p2/p1
(b) p3/p1 and
(c) T3/T1 For path 1 ?? 2, what are?
(d) W/nRT1,
(e) Q/nRT1,
(f) ΔEint/nRT1, and
(g) ΔS/nR? For path 2 ?? 3, what are?
(h) W/nRT1
(i) Q/nRT1,
(j) ΔEint/nRT1,
(k) ΔS/nR? For path 3 ?? L, what are?
(l) W/nRT1,
(m) Q/nRT1,
(n) ΔEint/nRT1,
(o) ΔS/nR?
Transcribed Image Text:
Isothermal Adiabatic Vas Volume Pressure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (18 reviews)
The connection between molar heat capacity and the degrees of freedom of a diatomic gas is given by setting f 5 in Eq 1951 Thus Cy5R2 C7R2 and y 75 In ...View the full answer

Answered By

Vineet Kumar Yadav
I am a biotech engineer and cleared jee exam 2 times and also i am a math tutor. topper comunity , chegg India, vedantu doubt expert( solving doubt for iit jee student on the online doubt solving app in live chat with student)
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Thermodynamics questions
-
A beat engine takes 0.350 mol of a diatomic ideal gas around the cycle shown in the pV-diagram of fig. 20.24. Process I 2 is at constant volume, process 23 is adiabatic, and process 3 1 is at a...
-
Two moles of a diatomic ideal gas are compressed isothermally from 18 L to 8 L. In the process, 170 calories escape from the system. Determine the amount of work done by the gas, the change in...
-
A 1.00-mol sample of a diatomic ideal gas has pressure P and volume V. When the gas is heated, its pressure triples and its volume doubles. This heating process includes two steps, the first at...
-
In the light of what you know about IHRM, how easy do you think it is to manage a global employer brand? What issues would an IHRM professional expect to have to manage? To answer this, analyse the...
-
Using the Stolper-Samuelson theorem, indicate the effect on the distribution of income between labor and capital in Nation 1 (assumed to be a small nation) when it imposes an import tariff on...
-
Investors have shown great interest in the stock of Rollover Beds Corporation, because the company has been growing at an average annual rate equal to 25 percent. Jason Jackson decided to evaluate...
-
What two fieldwork stages gather audit evidence?
-
Using first-in, first-out; perpetual inventory costing; and the following information, determine the cost of materials used and the cost of the July 31 inventory: July 1 Balance on hand, 1,000 yd of...
-
Could you help me with this finance question? AstroSnacks just paid a dividend of $2.00 per share, and the expected growth rate is 10%. If your required rate of return is 20%, what is the fair price...
-
Write a short proposal on the following topic. Class survey You have been studying English for a while now. Talk to other students and your teacher about the course and prepare a short proposal on...
-
At very low temperatures, the molar specific heat Cv of many solids is approximately Cv = AT3, where A depends on the particular substance. For aluminuffi, A = 3.15 X 10-5 J/mol K4. Find the entropy...
-
A 2.0 mol sample of an ideal monatomic gas undergoe the reversible process shown in Figure, the scale of the vertical axis is set by Ts = 400.0K and the scale of the horizontal axis is set by Ss =...
-
Vector A has y-component A y = +13.0 m. A makes an angle of 32.0 o counterclockwise from the y-axis. (a) What is the x-component of A? (b) What is the magnitude of A?
-
Check my work Max Company reports net income of $154,000 each year and declares an annual cash dividend of $40,000. The company holds net assets of $1,310,000 on January 1, 2023. On that date, Eden...
-
Discuss the role of symbolism and allegory in classic literature. How do these devices enhance the narrative's depth and invite diverse interpretations ?
-
Synthesize a statement of Georgia law regarding suppression of evidence, as applicable to the scenario presented below: The attorney you work for in Warner Robins, Georgia, has been hired to defend...
-
Sevenways, is a respected upper class residential complex in the so-called leafy surbubs of Nairobi. Kamau, an up-and-coming lawyer in Nairobi, was happy to buy a home for his family in this...
-
A block with a mass of 5 kg is placed on a inclined plane with an angle of inclination of 3 0 degrees. The coefficient of static friction between the block and the plane is 0 . 3 . a . Calculate the...
-
The circuit parameters of the Widlar current source in Figure 10.9 are \(V^{+}=3 \mathrm{~V}, V^{-}=-3 \mathrm{~V}\), and \(R_{1}=20 \mathrm{k} \Omega\). Assume \(V_{B E 1}(\mathrm{on})=0.7...
-
Estimate a range for the optimal objective value for the following LPs: (a) Minimize z = 5x1 + 2x2 Subject to X1 - x2 3 2x1 + 3x2 5 X1, x2 0 (b) Maximize z = x1 + 5x2 + 3x3 Subject to X1 + 2x2 +...
-
A 0.0250-kg bullet is accelerated from rest to a speed of 550 m/s in a 3.00-kg rifle. The pain of the rifles kick is much worse if you hold the gun loosely a few centimeters from your shoulder rather...
-
In Example 16.19 (Section 16.8), suppose the police car is moving away from the warehouse at 20 m/s. What frequency does the driver of the police car hear reflected from the warehouse?
-
Two train whistles, A and B, each have a frequency of 392 Hz. A is stationary and B is moving toward the right (away from A) at a speed of35.0 m/s. A listener is between the two whistles and is...
-
A railroad train is traveling at 25.0 m/s in still air. The frequency of the note emitted by the locomotive whistle is 400 Hz. What is the wavelength of the sound waves? (a) In front of the...
-
Please just draw the graph for the solution mentioned below: Thanks in advance! To calculate marginal cost (MC) and average cost (AC), we can use the given quadratic cost function. The formula f...
-
An entry to record Purchases and related Accounts Payable of $14,000 for merchandise purchased on December 23, 2026, was recorded in January 2027. This merchandise was not included in inventory at...
-
Learned Corporation has provided the following information: Cost per Unit Cost per Period Direct materials $ 5.00 Direct labor $ 3.60 Variable manufacturing overhead $ 1. 20 Fixed manufacturing...

Study smarter with the SolutionInn App


