Ketones undergo a reduction when treated with sodium borohydride, NaBH 4 . What is the structure of
Question:
Ketones undergo a reduction when treated with sodium borohydride, NaBH4. What is the structure of the compound produced by reaction of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm?1 arid M+ = 74 in the mass spectrum?
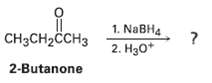
Transcribed Image Text:
1. NABH4 2. Hзо* |CH3CH2CCHЗ 2-Butanone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
CH3CH2CCH3 1 NaBH4 2 H3O OH ...View the full answer

Answered By

David Ngaruiya
i am a smart worker who concentrates on the content according to my clients' specifications and requirements.
4.50+
7+ Reviews
19+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the structure of ATCase?
-
What is the structure of the global beer industry?
-
What is the structure of the beer industry?
-
What do you think people would say about Corrie from the few quotes we have from her book? What was her personality like? Do you think she handled her incarceration differently than Elie Wiesel?...
-
Industries with high barriers to entry often have high barriers to exit. Explain.
-
Compute the level of on-balance volume (OBV) for the following 3-day period for a stock, if the beginning level of OBV is 50,000 and the stock closed yesterday at $25. Does the movement in OBV appear...
-
What is meant by the expectation gap?
-
Cleveland Area Rapid Delivery (CARD) operates a delivery service in the Cleveland met- ropolitan area. Most of CARDs business involves rapid delivery of documents and parcels between offices during...
-
Take some time to reflect on the key topics and concepts studied over the past ten weeks. Discuss two concepts or areas of knowledge that you feel have been enhanced for you during the course....
-
Fill out the market analysis worksheet based on the following information. The Subject property is located in Sweetbriar subdivision at 144 Sweetbriar Trail. It is an eight room, one story, wood...
-
Grignard reagents undergo a general and very useful reaction with ketones. Methyl magnesium bromide, for example, reacts with Cyclohexanone to yield a product with the formula C 7 H 14 O. What is the...
-
Nitriles, RC N, undergo a hydrolysis reaction when heated with aqueous acid. What is the structure of the compound produced by hydrolysis of propane nitrile, CH3CH2C N, if it has IR absorptions at...
-
(a) Suppose that we fix one node in a planar structure and put a second node on a roller. Does the structure admit any rigid motions? (b) How many rollers are needed to prevent all rigid motions in a...
-
Explain participative leadership style and give examples Explain what is meant by leadership style. Explain giving examples why greater attention has been given to more participative styles of...
-
Robertson Corporation acquired two inventory items at a lump-sum cost of $96,000. The acquisition included 3,000 units of product CF, and 7,000 units of product 3B. CF normally sells for $27 per...
-
The Narnia economy has Consumption Expenditures of $8,000; Exports of $6,000; Imports of $3,000; Investments of $6,000; inventories of $3,000; residential construction of $2,000; Total Government...
-
Express Delivery Company ( EDC ) is considering outsourcing its Payroll Department to a payroll processing company for an annual fee of $ 2 2 2 , 0 0 0 . An internally prepared report summarizes the...
-
Roberts Company is considering an investment in equipment that is capable of producing more efficiently than the current technology. The outlay required is $2,233,333.The equipment is expected to...
-
Consider the following tasks with their resource requirements given as: a) 1 = (10, 4, 1; [A; 1]), where the task executes for two time units, then requests the resource A. (b) 2 = (7, 4, 2; [A;...
-
a. What is the cost of borrowing if Amarjit borrows $28 500 and repays it over a four-year period? b. How many shares of each stock would he get if he used the $28 500 and invested equally in all...
-
The solubility of carbon tetrachloride (CCl 4 ) in water at 25 C is 1.2 g/L. The solubility of chloroform (CHCl 3 ) at the same temperature is 10.1 g/L. Why is chloroform almost ten times more...
-
Predict the products of the reactions of the following compounds with chromic acid and also with PCC. (a) Cyclohexanol (b) 1-methylcyclohexanol (c) Cyclopentylmethanol (d) Cyclohexanone (e)...
-
Two products are observed in the following reaction. (a) Suggest a mechanism to explain how these two products are formed. (b) Your mechanism for part (a) should be different from the usual mechanism...
-
Give the structures of the products you would expect when each alcohol reacts with (1) HCI, ZnCI2; HBr; (3) PBr3: (4) P/I2; (5) SOCI2- (a) butan-1-ol (b) 2-methylbutan-2-ol (c) 2,2-dimethylbutan-1-ol...
-
A stipulation from Senior Management that a design does not have to be fully complete before manufacturing some components can start is an example of a: a. crashing b. scope dependency c....
-
As the substitute for low - skill labor becomes available, the demand curve for low - skill labor will shift to the _ _ _ _ _ _ _ _ _ . Question 2 options: market; right market; left technology;...
-
In an income expenditure equilibrium: Group of answer choices there are no savings. there are no inventories. there is no unplanned inventory investment. inventory investment equals consumption.

Study smarter with the SolutionInn App


