Look at the following electrostatic potential map of chloromethane, and tell the direction of polarization of the
Question:
Look at the following electrostatic potential map of chloromethane, and tell the direction of polarization of the C ? C1 bonds:
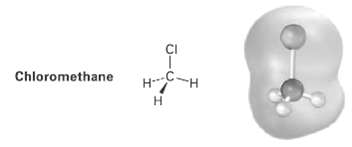
Transcribed Image Text:
CI C-H Chloromethane H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
In an electrostatic potential map the color red in...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following electrostatic potential diagrams represent H2, HCl, or NaCl. Label each, and explain your choices. (a) (b)
-
The following electrostatic potential diagrams represent CH4, NH3, or H2O. Label each, and explain your choices. a. C. b.
-
(a) Figure 11.16 is an electrostatic potential map of calicene, so named because its shape resembles a chalice (calix is the Latin word for "cup"). Both the electrostatic potential map and its...
-
A company operates a standard absorption costing system and absorbs fixed production overheads based on machine hours. The budgeted fixed production overheads for the company for the previous year...
-
Paul Barrone is a graduate student at State University. His 10-year-old son, Jamie, lives with him, and Paul is Jamie's sole support. Paul's wife died in 2015, and Paul has not remarried. Paul...
-
Who are the major players in a typical organized crime check fraud scheme?
-
On November 5, 2020, a Dunn Corporation truck was in an accident with an auto driven by R. Bell. Dunn received notice on January 12, 2021, of a lawsuit for \(\$ 350,000\) in damages for personal...
-
DepreciationStrike, Units-of-Production, Obsolescence Presented below and are three different and unrelated situations involving depreciation accounting. Answer the question(s) at the end of each...
-
Finlay works for Crystal Lighting in Alberta. Finlay earns an annual salary of $58,200.00 and is paid on a bi-weekly basis. She receives a $70.00 car allowance each pay. Crystal Lighting?...
-
Forty-two percent of adults say that they have cheated on a test or exam before. You randomly select six adults. Find the probability that the number of adults who say that they have cheated on a...
-
Use the electro-negatively values shown in Figure to rank the following bonds from least polar to most molar: H3C ? Li, H3C ? K, H3C ? F, H3C ? MgBr, H3C ? OH. 2.1 Li Be 1.0 1.6 Na Mg 0.9 1.2 Ca 0.8...
-
Ethylene glycol, HOCH2CH2OH, has zero dipole moment even though carbon-oxygen bonds are strongly polarized. Explain.
-
Construct an X bar-R Chart for the following data set. 1437 462437 12 60 02 170 04 14 96 18 04 66 12 92 2 1 2 233 233233232332 2340 1 .4 12 2 1 1 2 1 3 2 2 1 2 1 2 1 2 1 2 1 2 5 .4 4 4 4 1 2 031 0...
-
1. Dan is involved in three business activities. He does not materially participate in any activity. He has at-risk amounts in excess of $250,000 for each. During the year, the activities produced...
-
A company needs to raise 8M in equity from a venture capital fund to finance an expansion. The company will be worth as provided below in 5 years. If the venture capitalist wants to achieve a rate of...
-
1.17 A tension test performed on a metal specimen to fracture produced the stress-strain relationship shown in Figure P1.170. Graphically determine the following (show units and all work): 100 80...
-
During a penetration test, a security professional runs the following command during a security assessment: -(kali kali)-[~] nc -lvnp 8387 Ncat: Version 7.92 ( https://nmap.org/ncat ) Ncat: Listening...
-
A golfer hits a golf ball with a club head velocity of 81 ft/s. Mass of golf club head (my): 190 g Mass of golf ball (m): 46g Coefficient of restitution (COR) (e): 0.83 What is the velocity of the...
-
Consider the mass \(m\) suspended by a nonlinear hardening spring as shown in Figure 2.34. The spring obeys \(F=k x^{3}\) and has an original length of \(L\). The coordinate \(x\) is the stretch of...
-
Explain the circumstances that could result in a long-term bank loan being shown in a statement of financial position as a current liability.
-
The metal tin undergoes a transition from a gray phase to a white phase at 286 K and ambient pressure. Given that the enthalpy change of this transition is 2090 J/mol and that the volume change of...
-
Identify the most acidic hydrogen in each of these compounds: a) HOCCH,CH,SOH 0 CO H 0 I e) CHCCHCOCHCH3 0 b) CH-CH,CH,C=N d) 0 CHOH f) HNCHCOH
-
Show the products of these acid-base reactions and predict whether the equilibria favor the reactants or the products: a) CHCCHCCHCH + OCHCH b) CHCHNO + CHO: CH3 (c) CH3COCH, + CHCH- 10 1:Z: LL CH,...
-
Which compound is behaving as the Lewis acid and which as the Lewis base in this reaction? AICI3 T CHCHCHCH3 + AICI CHCHCHCH3
-
This homework is a mathematically oriented assignment designed to equip you with some of the techniques we'll need going forward to describe robotic systems. In particular, we'll focus on the...
-
Item 5 (6 points) List three email protocols from Chapter 1: Email protocol Does it Send or Receive email? Feature Email is stored until a client requests delivery
-
Choose the correct description of the population. A. The income of home owners in the country who work at home B. The number of home owners in the country who work at home C. The income of home...

Study smarter with the SolutionInn App


