Propose a structure for a compound C4H7N that has the following IR and 1H NMR spectra: 100
Question:
Propose a structure for a compound C4H7N that has the following IR and 1H NMR spectra:
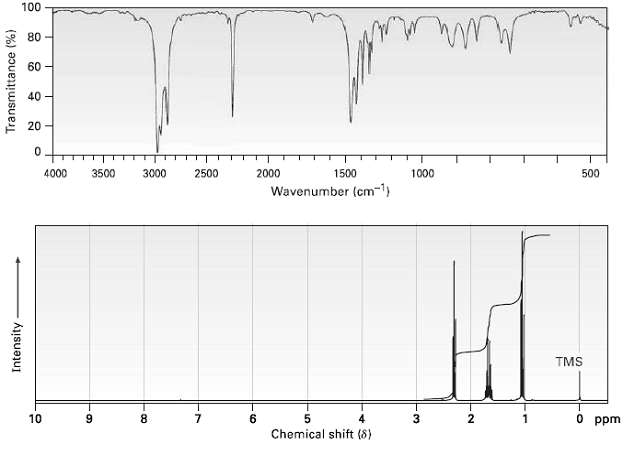
Transcribed Image Text:
100 80 - 60 40 - 20 - 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm-1) TMS O ppm 10 9. 8. 3. Chemical shift (8) Transmittance (%) Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
A compound with the formula C4HN ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The IR and 1H NMR spectra of phenacetin (C10H13NO2) are given in Fig. 17.7. Phenacetin is an analgesic and antipyretic compound and was the P of A-P-C tablets (aspirin-phenacetin-caffeine). (Because...
-
Compound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and assign each peak in the NMR spectrum. Note that the absorption at 5.5 ?...
-
A compound (C8H10O) has the infrared and 1H NMR spectra presented in Figure 13.42. What is its structure? Figure 13.42 Wave number, cm 5.0 48 Ppm) .60 PPm 10.0 9.0 8.0 7.0 6.05.0 4,0 3.0 2.0 .0 O...
-
Read the Speech of Martin Luther King, Jr.'s "I Have a Dream" and answer the question: Kin, makes liberal use of metaphorand metaphorical imagery in his speech. (Glossary: Figures of Speech) Choose a...
-
1. How should Wes and his team go about developing a survival stratgegy for Saratoga Supply? 2. Does Sarataoga Supply really need a survival strategy? Or should the company just begting to downsize...
-
A soft-drink company produces 100,000 12-ounce bottles of soda per year. By adjusting a dial, the company can set the mean number of ounces placed in a bottle. Regardless of the mean, the standard...
-
Stonewall Corporation issued \(\$ 20,000\) of \(5 \%, 10\)-year convertible bonds. Each \(\$ 1,000\) bond is convertible to 10 shares of common stock (par \$50) of Stonewall Corporation. The bonds...
-
Lynn Rogers (who just turned 30) currently earns $60,000 per year. At the end of each calendar year, she plans to invest 10% of her annual income in a tax-deferred retirement account. Lynn expects...
-
Suppose that the current 1-year rate (1-year spot rate) andexpected 1-year T-bill rates over the following three years (i.e.,years 2, 3, and 4, respectively) are as follows:1R1 = 3.10%,E(2r1) = 4 2...
-
Waldrup Industries is considering a proposal for a joint venture that will require an investment of C$13 million. At the end of the fifth year, Waldrup's joint venture partner will buy out Waldrup's...
-
Compound A, C 4 H 8 O 3 , has infrared absorptions at 1710 and 2500 to 3100 cm ?1 and has the 1 H NMR spectrum shown. Propose a structure for A. 11.18 TMS 10 O ppm 8. 6. 3. Chemical shift (6)...
-
The two 1H NMR spectra shown here belong to crotonic acid (trans-CH3CH = CHCO2H) and meth acrylic acid [H2C = C (CH3) CO2H]. Which spectrum corresponds to which acid? Explain. (a) 12.2 8 TMS O ppm 10...
-
For each given slope, identify the line in AD that could have this slope. -3 A. B. C. D.
-
Hudson Company reports the following contribution margin income statement. HUDSON COMPANY Contribution Margin Income Statement For Year Ended December 31 Sales (11,400 units at $225 each) Variable...
-
-4 -x+4y+32 Consider the 3 x 3 matrix A= 3 3x-12y-10z 3 -11 2x-8y-7z -8 -1 Find all the possible values of x, y and z for which A is symmetric.
-
If you were in Genpact s human resource department, would you recommend that the company roll out the same kind of performance feedback to the remaining teams? Why or why not?
-
Give the name of the case at 555 U.S. 223 (2009). b. Briefly describe the general topic of this case. c. What was the holding in this case? d. What was the "vote" in this case? 5. Locate and review...
-
1) Why do you think Colin Powell identified trust as one of the most important things leaders need to work on with their teams? 2) Identify and provide rationale for two other traits you feel are...
-
Refer to Problem 5. Find the mean number of pets. Problem 5 The following table presents a probability distribution for the number of pets each family has in a certain neighborhood. Construct a...
-
For the following arrangements, discuss whether they are 'in substance' lease transactions, and thus fall under the ambit of IAS 17.
-
Calculate the packing efficiency of the face-centered cubic unit cell. Show your work.
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
Identify the etlryl groups and the methyl groups in the structure of 4-sec-butyl-5ethyl-3- methyloctane, the compound discussed in Study problem 2.5. Note that these groups are not necessarily...
-
Identify the etlryl groups and the methyl groups in the structure of 4-sec-butyl-5ethyl-3- methyloctane, the compound discussed in Study problem 2.5. Note that these groups are not necessarily...
-
Problem 1-18 Filling Status and Tax Computation, Qualifying Dependents (LO 1.5, 1.6) Karl is a single taxpayer and has listed his brother, Jonas, as a dependent (qualifying relative). Head of...
-
Another flaw in the design of the building resulted in an extreme amount of swaying. In fact, office workers in the upper stories of the building complained of motion sickness. The building actually...
-
Below are 3 scatterplots. Which of the datasets has the strongest correlation. C OB OA B d A

Study smarter with the SolutionInn App


