Rank the following substances in order of their expected SN1 reactivity: Br Br H CHCCH CHCH2Br H2C=CHBr
Question:
Rank the following substances in order of their expected SN1 reactivity:
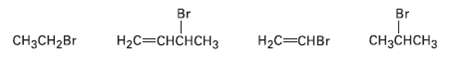
Transcribed Image Text:
Br Br Нас—снснсHз CHзCнCHз CHзCH2Br H2C=CHBr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Syl reactivity is related to carbocation ...View the full answer

Answered By

Gaurav Soni
Teaching was always an area where I can pursue my passion. I used to teach my friends and junior during my school and college life. After completing my professional qualification (chartered accountancy) and before joining my job, I also joined an organization for teaching and guidance to my juniors. I had also written some articles during my internship which later got published. apart from that, I have also given some presentations on certain amendments/complex issues in various forms.
Linkedin profile link:
https://www.linkedin.com/in/gaurav-soni-38067110a
5.00+
7+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following substances in order of increasing acidity: C CH3CH3 CHCCH2CCH Acetic acid (pKa = 4.76) Phenol Acetone (pKa = 19.3) 2,4-Pentanedione (pKa = 9) (pKa = 9.9) %3D %3D
-
Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC CH, (CF3)2CHOH, CH4OH (b) Phenol, p-methyl phenol, p-(trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxy...
-
List the following substances in order of their increasing oxidizing power: (a) Acetoacetate, (b) Cytochrome b (Fe3+), (c) NAD+ (d) SO24-, and (e) Pyruvate.
-
Umida Ltd is considering acquiring Trinity Ltd. Both companies are all-equity firms. Umida and Trinity have 5 million and 6 million shares outstanding respectively. Umida generates $2 million in...
-
Based on the information given, which sources of job satisfaction has Datotel addressed? What other sources might the company address, and how?
-
A result from First Corporations most recent year of operations is presented in the following table. Operating income....................................................................................
-
The company had one temporary difference due to the GAAP basis of equipment exceeding the tax basis of the equipment. Record the income tax journal entry for 2020, assuming an enacted tax rate of...
-
Operating Transactions, Special Topics, and Financial Statements. The City of Ashland's General Fund had the following post-closing trial balance at April 30, 2010, the end of its fiscal year: During...
-
taxpayer purchased an annuity from an insurance company that promised to pay her $2,100 per month for 15 years. Taxpayer paid a premium of $272,160 for the annuity. Determine the amount of the first...
-
Court Opinion 4-1 United States V. Yoshida, 303 F.3d 1145 (9th Cir. 2002) Section 1324(a)(1)(A)(iv) makes it a crime to (1) encourage or induce, (2) an alien to come to the United States, (3) while...
-
Assign configuration to the following substrate, and show the stereo chemistry and identity of the product you would obtain by SN1 reaction with water (reddish brown =Br):
-
3-Bromo-1-butene and 1-bromo-2-butene undergo SN1 reaction at nearly the same rate even though one is a secondary halide and the other is primary. Explain
-
Identify each of the following items relating to sections of an income statement as Revenue from Sales (S), Cost of Goods Sold (CGS), Selling Expenses (SE), General Expenses (GE), Other Income (OI),...
-
You are the general manager of a home-mortgage lending business within a large diversified financial services firm. In the firm's mission statement, there is a value that emphasizes the importance of...
-
Describe a sociology concept or theory that clarifies why academic dishonesty is increasing among post-secondarystudentsand offers apreventativemeasure. THE ESSAY INCLUDES: INTRODUCTION - present the...
-
How to differentiate between the two accounting concepts: cash basis accounting and accrual basis of accounting ?
-
Air at 6 0 \\ deg C contains 2 5 g / m ? of water vapor. ( a ) What is the relative humidity? ( b ) What is the dew point? ( c ) What is the wet - bulb temperature. The answer for a = 1 9 . 1 % , b =...
-
Thoroughly explain the interest group model of regulation. Provide peer-reviewed journal reference along with a Biblical integration detailing scripture or a Christian worldview.?
-
Again consider the three temperature series of Europe, North America, and South America. Build a vector ARMA model (AR or MA model is allowed) for the three series. Perform model checking. Obtain...
-
Keating & Partners is a law firm specializing in labour relations and employee-related work. It employs 25 professionals (5 partners and 20 managers) who work directly with its clients. The average...
-
Coal, which is primarily carbon, can be converted to natural gas, primarily CH 4 , by the exothermic reaction: Which disturbance will favor CH 4 at equilibrium? a. Adding more C to the reaction...
-
The following partial IR spectra correspond to a primary amine, a secondary amine, and an alcohol. Give the functional group for each spectrum. 2.5 100 3 3.5 4 3 4 2.5 100 3.5 4 100 (by 3500 4000...
-
The proton and 13C NMR spectra of a compound of formula C4H11N are shown here. Determine the structure of this amine, and give peak assignments for all of the protons in the structure. 200 180 160...
-
The carbon NMR chemical shifts of diethylmethylamine, piperidine, propan-1-ol, and propanal follow. Determine which spectrum corresponds to each structure, and show which carbon atom(s) are...
-
Costs incurred in the process of making products, including direct materials, direct labor and manufacturing overhead are _blank_ costs. Multiple choice question. upstream downstream midstream
-
true or false 4. The expenditure of $30 million cash to obtain station rights for the next five years at Reagan National airport is a cash outflow related to operating activities of the business....
-
The DEF Co . has planned the following sales for the next three months: Jan Feb Mar Budgeted sales $ 4 0 , 0 0 0 $ 5 0 , 0 0 0 $ 7 0 , 0 0 0 Sales are made 2 0 % for cash and 8 0 % on account. From...

Study smarter with the SolutionInn App


