Show the products of thesereactions: 1) Hg(O,CCH,) . 2) NaBH3. NAOH 1) Hg(O,CCH,)2, H,0 2) NaBH4, NaOH
Question:
Show the products of thesereactions:
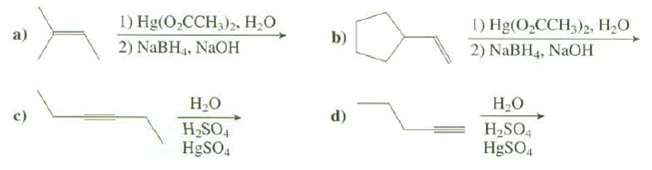
Transcribed Image Text:
1) Hg(O,CCH,)» Н.О 2) NaBH3. NAOH 1) Hg(O,CCH,)2, H,0 2) NaBH4, NaOH b) a) Н.о H SO4 Н.о H2SO4 d) HgSO, H9SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (12 reviews)
a...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Calculate the density of N2 at STP, (a) using the ideal-gas law and (b) using the molar volume and molar mass of N2. How do the densities compare?
-
Wilson revises her estimates of the benefits from the new system's lower labor costs as calculated in P1-28A. She now thinks the savings will be only $935,000. Requirements 1. Compute the expected...
-
Your firm has done the audit of Rhea Fashions Inc. for many years. You are in charge of the fieldwork for the current years audit. Rhea is a manufacturer of high-fashion clothing. Its shares are...
-
Find positive weights \(w\) in a utility function of the form \(u\left(c_{1}, c_{2} ight)=\) \(c_{1}+w \cdot c_{2}\) to create at least four different strict rankings among the candidates \(x, y, z\)...
-
Dollar-Value LIFO The following information relates to the Choctaw Company. Use the dollar-value LIFO method to compute the ending inventory for Choctaw Company for 2007 through 2011. Ending...
-
Round 3. 7. Suppose that the mean number of fatal car accidents each year in Columbus is 26.5 and these accidents follow a Poisson distribution. a) Find the mean and standard deviation of the number...
-
A Global private bank is aggressively looking to leverage technology to improve customer experience and reduce operational costs. Over the last few years, it has tied up with at least five startups...
-
Show all the steps in the mechanism for the formation of MTBE from methanol and isobutylene.
-
Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
-
Explain when a firm should use each of four major ways to enter a foreign market.
-
Differentiate between botany and zoology.? Name sub branches with example of each
-
Some argue that certain HR practices create sustained competitive advantage (best practices approach) while others argue that what is more important in the match between HR practices and other...
-
Hans Olo Bros. has developed a new product that will be marketed for the first time next year. Although the marketing department estimates that 35,000 units could be sold at $36 per unit, Hans Olos...
-
As an HR Manager, what changes do you think should be implemented to increase the productivity of your department? What are the three standards developed by the American Psychological Association for...
-
As of December 3 1 , 2 0 2 2 , Stand Still Industries had $ 7 0 0 of raw materials inventory. At the beginning of 2 0 2 2 , there was $ 3 , 0 0 0 of materials on hand. During the year, the company...
-
The external and internal radii of a friction plate of a clutch are \(120 \mathrm{~mm}\) and \(60 \mathrm{~mm}\) respectively. The total axial thrust is \(1500 \mathrm{~N}\). For uniform wear, find...
-
4. Jobe dy -Y 2 et by
-
Calculate the value of the equilibrium constant for each of the following reactions in aqueous solution. a. HCHO + OHCHO + HO b. C,H,O, +H*
-
Predict the product(s) of the following reactions. If more than one product is formed, tell which ismajor. (a) CH3I (excess) Ag20, H20 eat C? A? B? .cocI (b) H20 NaN3 eat C? B? A? (c) CgHsCH2Br C7...
-
Fill in the missing reagents a?c in the following scheme: NH2 CHCH3 CH=CH2 CCH3 b, c CHCH2NCH3 CH CH-CH2
-
Although pyrrole is a much weaker base than most other amines, it is a much stronger acid (pK a 15 for the pyrrole versus 35 for diethyl amine). The NH proton is readily abstracted by base to yield...
-
Based on my part 1, I need help doing the second part. You will research and identify the data I would need to manage as part of my application. Determine tables and columns required for my...
-
. The table given below shows how, on average, the market value of a Boeing 737 has varied with its age and the cash flow needed in each year to provide a 13%% return. (For example. if you bought a...
-
vas Gateway (LO2) Sprinkle Co. sells its product for $20 per unit. During 2013, it produced 60,000 units and sold 50,000 units (there was no beginning inventory). Costs per unit are: direct materials...

Study smarter with the SolutionInn App


