Look at this paper chromatogram: a. The solvent used was ethanol. Which sample of ink, A, B
Question:
Look at this paper chromatogram:
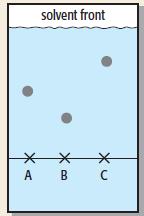
a. The solvent used was ethanol. Which sample of ink, A, B or C, has the greatest relative solubility in ethanol?
b. Work out the Rf value of the ink whose partition coefficient in ethanol and water lies between the values of the other two inks.
Transcribed Image Text:
solvent front A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
a First we note that the paper chromatogram is a mixture of just two components ink ...View the full answer

Answered By

Enock Oduor
I am a chemist by profession, i coach high school students with their homework, i also do more research during my free time, i attend educational and science fair seminars where i meet students and do some projects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Question Posted:
Students also viewed these Sciences questions
-
A box is separated by a partition into two parts of equal volume. The left side of the box contains 500 molecules of nitrogen gas; the right side contains 100 molecules of oxygen gas. The two gases...
-
In Figure, a bar magnet lies near a paper cylinder. (a) Sketch the magnetic field lines that pass through the surface of the cylinder. (b) What is the sign of B ?? d A for every area d A on the...
-
Ink Spot is a small company that has been in business for two years. Wilford Smith, the president of the company, has decided that it is time to expand. He needs $10,000 to purchase additional...
-
This can be anything - goods or services; in which you reflect upon your understanding of the ideal marketing mix for your proposed product/service. please explain for me in details and sources?
-
You have two investment alternatives. Alternative 1 requires an immediate outlay of $8000. In return, you will receive $900 at the end of every quarter for the next three years. Alternative 2...
-
Okamura Construction Corp. has a defined benefit From the Plan Actuary: - Current service cost in \(20 \mathrm{X} 7\) is \(\$ 430,000\) and in \(20 X 8\) is \(\$ 488,000\). - Defined benefit...
-
Security Atlantic Bank Corporation is evaluating two capital investment proposals for a drive-up ATM, each requiring an investment of \($200,000\) and each with an 8-year life and expected total net...
-
What is a major difference between iGAAP and U.S. GAAP as regards revenue recognition practices?
-
Was the intervention effective for each participant? Provide a rationale. Make sure that you discuss the adequacy of the number of data points in each phase, level, trend, variability, consistency,...
-
For this exercise, your client, Bright IDEAs Inc., has provided you with data for two related files, a listing of sales invoices, and a listing of customers with credit limits. To test whether credit...
-
The gasliquid chromatogram for a mixture of organic compounds is shown below. a. Give the correct labels for a, B and C. b. What percentage of the mixture is pentan-1-ol? c. Give an explanation for...
-
a. Identify the fragments that would cause peaks in the mass spectrum of HOCH 2 COCH 3 with the following m/e values: i. m/e = 15 ii. m/e = 17 iii. m/e = 31 iv. m/e = 43 v. m/e = 57 vi. m/e = 59 b....
-
The end area at station 36 + 00 is 2 265 ft . Notes giving distance from centerline and cut ordinates for station 36 + 60 are C 4.8/17.2; C 5.9/0; C 6.8/20.2. Base is 20 ft.
-
The following are the transactions for the month of July. July 1 July 13 Beginning Inventory Purchase Units 58 Unit Cost $ 10 Unit Selling Price 290 11 July 25 July 31 Sold Ending Inventory (100) 248...
-
Assume the group, in whole or in part, is able to proceed with the hotel development project, and a possible site has been identified for the first hotel. You set up a conference call with the group,...
-
Provide a summary of different Ipv6 routing protocols. How are they similar to IPv4 protocols? How are they different? What is SLAAC (Stateless Address Auto Configuration)? How does it work? What are...
-
Yost received 300 NQOs (each option gives Yost the right to purchase 10 shares of Cutter Corporation stock for $15 per share). At the time he started working for Cutter Corporation three years ago,...
-
Below are amounts (in millions) from three companies' annual reports. Beginning Accounts Receivable Ending Accounts Receivable $2,692 Net Sales $315,427 WalCo TarMart CostGet Required: $1,745 5,816...
-
What are the advantages of the withdrawal rate method?
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
Is the following statement correct? If not rewrite it so that it is correct. The superscript zero in H o f means that the reactions conditions are 298.15 K.
-
Why is it valid to add the enthalpies of any sequence of reactions to obtain the enthalpy of the reaction that is the sum of the individual reactions?
-
Identify the expected hybridization state and geometry for the central atom in each of the following compounds: a. b. c. d. e. H. I-z: I-
-
Garcia Industries uses a cost system that carries direct materials inventory at a standard cost. The controller has established these standards for the cost of one unit: Standard Quantity X Standard...
-
In a small town of 5,832 people, the mayor claims that there is a difference in the proportion of voters ages 18-30 who would support an increase in the food tax and the proportion of voters ages...
-
Suppose that the continuously compounding annualized interest rate is 10% and that the price of a S&R stock is $1,000. a. Suppose that you are offered a one-year long forward contract on S&R stocks...

Study smarter with the SolutionInn App


