Calculate the standard heats and Gibbs energies of reaction at 25C for the following reactions: a. N(g)
Question:
Calculate the standard heats and Gibbs energies of reaction at 25°C for the following reactions:
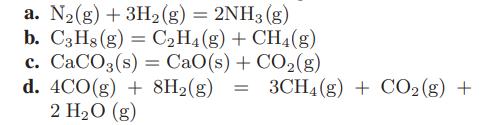
Transcribed Image Text:
a. N₂(g) + 3H₂(g) = 2NH3 (g) b. C3H8 (g) = C₂H4 (g) + CH4(g) c. CaCO3(s) = CaO(s) + CO₂(g) d. 4CO(g) + 8H2(g) = 3CH4(g) + CO₂(g) + 2 H₂O (g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To calculate the standard heats AH and Gibbs energies AG of reaction at 25C for the given reactions ...View the full answer

Answered By

Caroline Kinuthia
Taking care of the smaller details in life has a larger impact in our general well being, and that is what i believe in. My name is Carol. Writing is my passion. To me, doing a task is one thing, and delivering results from the task is another thing. I am a perfectionist who always take things seriously and deliver to the best of my knowledge.
4.90+
1934+ Reviews
4273+ Question Solved
Related Book For 

Chemical Biochemical And Engineering Thermodynamics
ISBN: 9780470504796
5th Edition
Authors: Stanley I. Sandler
Question Posted:
Students also viewed these Engineering questions
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Fe3 + (aq) + Cu(s) Fe2+(aq) + Cu2+(aq) The equation is not balanced.
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Sn4+(aq) + 2Hg(l Sn2+(aq) + Hg2 2+(aq) The equation is not balanced.
-
Calculate the standard free-energy change at 25C for the following reaction. 4Al(s) + 3O2(g) + 12H + (aq) 4Al3 + (aq) + 6H2O(l) Use standard electrode potentials.
-
Mookie The Beagle Concierge Trial Balance As of January 31,2023 is given 1001 Checking 1010 Money Market 1100 Accounts Receivable (A/R) 1210 Prepaid Expenses:Supplies 1220 Prepaid Expenses:Insurance...
-
Alex is a wage earner with a regular income. He has unsecured debts of $42,000 and secured debts owing to Betty, Connie, David, and Eunice totaling $120,000. Eunices debt is secured only by a...
-
Suppose you discovered that on your college campus, 6% of the female students were married and 4% of the female students had at least one child. a. From this information, is it possible to determine...
-
Compute output in terms of equivalent units.
-
Three different companies each purchased a machine on January 1, 2013, for $64,000. Each machine was expected to last five years or 200,000 hours. Salvage value was estimated to be $4,000. All three...
-
Perform a capital budgeting analysis for Microsoft Corporation's investment in a new software development project. Calculate the net present value (NPV), internal rate of return (IRR), and payback...
-
Mr. M has been employed as an engineer by A Ltd., a company incorporated in Hong Kong. During the year ended 31 March 2019, Mr. M had the following income and expenditure. A monthly salary of...
-
Calculate the minimum work required to separate air (79 mole % nitrogen) into pure oxygen and nitrogen assuming an isothermal, steady flow process at 300 K. The inlet air pressure is 10 bar and each...
-
a. Given experimental data either for the excess Gibbs energy, G ex , or for species activity coefficients from which G ex can be computed, it is sometimes difficult to decide whether to fit the data...
-
In a sample of 158 cartridges from a certain lot, 36 were found to be contaminated and 122 were "clean." If you randomly select 5 of these 158 cartridges, what is the probability that all 5 will be...
-
Current dividend $7.80, years until dividend ceases 13, required return 11.2%. Calculate the share price
-
In the figure below, two elastic members AB and CD are of rectangular cross section and have the same cross-sectional area A. They are made of the same material with Young's modulus E. They are pined...
-
Consider the following figure. 8.00 C + y 2.00 . 4.00 C @ (a) Find the electric potential, taking zero at infinity, at the upper right corner (the corner without a charge) of the rectangle in the...
-
Silven Industries, which manufactures and sells a highly successful line of summer lotions and insect repellents, has decided to diversify in order to stabilize sales throughout the year. A natural...
-
In more simplified format and at your own words advise What are the benefits of category management?
-
Identify and briefly explain the four variables or characteristics for describing a target market.
-
If your school has a subscription to the FASB Codification, go to aaahq.org/ ascLogin.cfm to log in and prepare responses to the following. (a) What is the stock dividend? (b) What is a stock split?...
-
Use MATLAB to calculate a. b. c. d. Check your answers with a calculator. el-2.1 + 3.47 log(14) + V287
-
Use MATLAB to calculate a. 6 tan -1 (12.5) + 4 b. 5 tan [3 sin -1 (13/5)] c. 5 ln(7) d. 5 log(7) Check your answers with a calculator.
-
The Richter scale is a measure of the intensity of an earthquake. The energy E (in joules) released by the quake is related to the magnitude M on the Richter scale as follows. E = 10 4.4 10 1.5M How...
-
Required: Following Simplicity Financial and Business Advisory Group Pty Ltd policies and procedures prepare the following: Task 1 Financial statement analysis The client needs a review of financial...
-
SCC Company reported the following for the current year.Net sales$ 57,000Cost of goods sold47, 200Beginning balance in inventory2,900Ending balance in inventory8,960Compute (a) inventory turnover and...
-
You are an assistant financial consultant for Simplicity Financial and Business Advisory Group PtyLtd, and you have been notified the client converted their business to a public company PTCG Ltd....

Study smarter with the SolutionInn App


