Name the following compounds. a. b. c. CH3, H C=C Br H
Question:
Name the following compounds.
a.
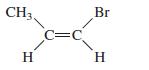
b.
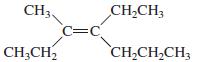
c.
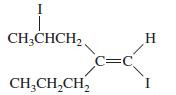
Transcribed Image Text:
CH3, H C=C Br H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The images provided show the structures of three organic compounds each containing a double bond indicating that they are alkenes I will name each com...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
ww 1. Two years ago the rental for the use of equipment and facilities was paid 5 years in advance with option to renew the rent for another 5 years by payment of P15,000 annually at the start of of...
-
Name the following compounds by IUPAC rules: a. b. H-C CH,CH-CH
-
Name the following compounds by the IUPAC system: a. CH3CH=C(CH2CH2CH3)2 b. (CH3)2CHCH"CHCH3 c. g. CH3-C-C-CH-CH, h. k.
-
A student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all the NBS has reacted. After a careful distillation, the product mixture contains two major...
-
Use Part 1 of the Fundamental Theorem of Calculus to find the derivative of the function. 8. g(x) = (" In t dt 7. g(x) = VT+ 21 dt sin t dt dx 10. glu) = 9. g(y) 11. F(x) = cos(r) dt Hint: [" costr?)...
-
Make the given changes in the indicated examples of this section and then factor. In Example 1, change the 3 to 4 and the 2 to 3. Data from Example 1 In factoring x 2 + 3x + 2, we set it up as The...
-
Net sales, net income, and total asset figures for Janice Controls, Inc., for five consecutive years are given below (Janice manufactures pollution controls): Required a. Calculate trend percentages,...
-
At the start of the current year, Blue Corporation (a calendar year taxpayer) has accumulated E & P of $100,000. Blue's current E & P is $60,000, and at the end of the year, it distributes $200,000...
-
Which type of mutual fund is most likely to have the lowest investment management fees? a. A bond fund. b. An equity growth fund. c. A small cap fund. d. An index fund.
-
Minoxidil \(\left(\mathrm{C}_{9} \mathrm{H}_{15} \mathrm{~N}_{5} \mathrm{O}ight)\) is a compound produced by the Pharmacia \& Upjohn Company that has been approved as a treatment for some types of...
-
Draw the following. a. cis-2-hexene b. trans-2-butene c. cis-2,3-dichloro-2-pentene
-
Pyridine is a weak organic base and readily forms a salt with hydrochloric acid. What is the pH of a 0.025 M solution of pyridinium hydrochloride, [C 5 H 5 NH + ]Cl ? CH5N(aq) + HCl(aq) C5H5NH*(aq)...
-
When you 'pop' popcorn, you are essentially conducting an experiment using Gay- Lussac's Law (there are a couple of other things going on here, too, but let's just focus on the gas laws). There is a...
-
Use the same circuit as part (a) (repeated below). Assuming the diode has a constant voltage drop of 0.7V, sketch Vout versus time for the triangular wave input shown below, and answer the questions....
-
Although it seems like women-owned and veteran women-owned business are in the rise, it is still very challenging for women to get the capital needed for their business. Investors will give the...
-
How does functionalism conceptualize the relationship between religion and society, exploring how religious beliefs, rituals, and institutions serve functional purposes in promoting social cohesion,...
-
For each of the following systems, determine whether they are static or dynamic linear or non-linear ii) v) time-invariant or time-varying causal or non-causal Stable or unstable. Hints: a. if you...
-
An electronics company makes communications devices for military contracts. The company just completed two contracts. The navy contract was for 2,300 devices and took 25 workers two weeks (40 hours...
-
10m solution. If Ka(HA) = 10 then pOH of solution will be [Given : log4=0.6] (A) 6.7 (B) Greater than 6.7 & less than 7.0 (C) Greater 7.0 & less than 7.3 (D) Greater than 7.3
-
Adenosine triphosphate (ATP) is a compound that provides energy for biochemical reactions in the body when it undergoes hydrolysis. For the hydrolysis of ATP at 37C (normal body temperature), H r =...
-
(a) In an experiment, 5.0 mmol Cl 2 (g) was sealed into a reaction vessel of volume 2.0 L and heated to 1200. K, and the dissociation equilibrium was established. What is the equilibrium composition...
-
The following groups are found in some organic molecules. Which are hydrophilic and which are hydrophobic: (a) OH; (b) CH 2 CH 3 ; (c) CONH 2 ; (d) Cl?
-
A study was conducted in a grocery store. Shoppers were given samples of fruit jams in two different situations: In the "limited choice" situation they were able to choose from six flavors, while in...
-
The military contractor mentioned earlier decided to do a 3 month weighted moving average. The weights are 60% for the most recent, 30 percent for the middle period and 10% for the oldest data. This...
-
Demand for 64 oz. orange juice cartons at a local supermarket is constant at the rate of 2000 cartons per month. The supermarket can purchase the 64 oz. cartons from supplier A at a price of...

Study smarter with the SolutionInn App


