The decomposition of N 2 O 5 in the gas phase was studied at constant temperature: The
Question:
The decomposition of N2O5 in the gas phase was studied at constant temperature:
![]()
The following results were collected:
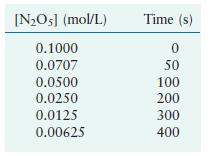
Using these data, verify that the rate law is first order in [N2O5], and calculate the value of the rate constant, where the rate = –d[N2O5]/dt.
Transcribed Image Text:
2NOs(g) 4NO(g) + O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
We can verify that the rate law is first order in NO5 by ...View the full answer

Answered By

Muhammad Salman Alvi
Well, I am a student of Electrical Engineeing from Information Technology University of Punjab. Just getting into my final year. I have always been good at doing Mathematics, Physics, hardware and technical subjects. Teaching profession requires a alot of responsibilities and challenges.
My teaching experience started as an home tutor a year ago. When I started teaching mathematics and physic subjects to an O Level student. He was about 14 years old. His name was Ibrahim and I used to teach him for about 2 hours daily. Teaching him required a lot of patience but I had to be polite with him. I used to give him a 5 min break after 1 hour session. He was quite weak in basic maths and calculation. He used to do quite a lot of mistakes in his homework which I gave him weekly. So I decided to teach him basics from scratch. He used to say that he got the concept even if he didn't. So I had to ask him again and again. I worked on his basics for a month and after that I started taking a weekly test sesions. After few months he started to improve gradually. Now after teaching him for about a year I can proudly say that he has improved alot. The most important thing was he managed to communicate all the difficullties he was facing. He was quite capable and patient. I had a sincere desire to help him reach to its full potential. So I managed to do that. We had a very good honest relationship of a student and a teacher. I loved teaching him as a tutor. Now having an experience of one year teaching I can read students quite well. I look forward to work as an online tutor who could help students in solving their all sort of difficulties, problems and queries.
4.90+
29+ Reviews
43+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using the data given in Example 15.2, calculate [N 2 O 5 ] 150. s after the start of the reaction. Data from Example 15.2 The decomposition of N 2 O 5 in the gas phase was studied at constant...
-
The decomposition reaction of N2O5 in carbon tetrachloride is 2 N2O5 -- 4 NO2 + O2. The rate law is first order in N2O5. At the rate constant is 4.82 10-3 s-1. (a) Write the rate law for the...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Use the DerivaGem software to value a five-year collar that guarantees that the maximum and minimum interest rates on a LIBOR-based loan (with quarterly resets) are 7% and 5% respectively. The LIBOR...
-
(a) Solve the differential equation y = 2x1 y2. (b) Solve the initial-value problem y = 2x1 y2, y (0) = 0, and graph the solution. (c) Does the initial-value problem y = 2x1 y2, y (0) = 2, have a...
-
Answer the following questions and discuss them in class. Explain the differences between a limited partnership and other partnerships
-
Part One: Measuring Efficiency and Effectiveness of Discretionary Cost My Fitness Inc., an athletic club, hires athletic trainers at its facilities primarily to offer individualized training sessions...
-
Jeffrey Helm owns a health and fitness center called Bulk- Up in Harrisburg. He is considering adding more floor space to meet increasing demand. He will either add no floor space ( N), a moderate...
-
(a) State any two functions of the financial system in any economy. (b) What is credit risk? (e) What is high powered money? (4) Distinguish between primary and secondary market. (e) What is risk...
-
A certain first-order reaction has a half-life of 20.0 minutes. a. Calculate the rate constant for this reaction. b. How much time is required for this reaction to be 75% complete?
-
The reaction between bromate ions and bromide ions in acidic aqueous solution is given by the following equation: Table 15.5 gives the results of four experiments involving this reaction. Using these...
-
1. What strategy has James Dyson used to develop and sell products like his cyclonic vacuum cleaner? 2. What leadership qualities does James Dyson exhibit? 3. What do you think is the Dyson Companys...
-
The transactions of Spade Company appear below. a. K. Spade, owner, invested $19,000 cash in the company in exchange for common stock. b. The company purchased supplies for $551 cash. c. The company...
-
Determine the implied domain of the following function. Express your answer in interval notation. Answer f(x) = = x+7 9
-
After watching the video from "The Big Bang Theory, describe what was occurring in the scene. What types of listening were demonstrated. How could this conversation be improved so that both parties...
-
Discuss the barriers to effective communication. How can these barriers be overcome by managers, and why is doing so important?
-
Ryan wants to show the height of a cliff. He is standing at the top of a cliff. He says that he can figure out how high the cliff is by throwing a rock. If he throws the rock with a horizontal...
-
The following table gives the operation times and due dates for i ve jobs that are to be processed on a machine. Assign the jobs according to the shortest operation time and calculate the mean...
-
State whether each of the following will increase or decrease the power of a one-way between-subjects ANOVA. (a) The effect size increases. (b) Mean square error decreases. (c) Mean square between...
-
Draw the Lewis structure and predict the shape of (a) OSbCl 3 ; (b) SO 2 Cl 2 ; (c) IO2F 2 . The atom in boldface red type is the central atom.
-
Structural isomers are molecules that have the same composition but a different pattern of connectivity. Two isomers of disulfur difluoride, S 2 F 2 , are known. In each, the two S atoms are bonded...
-
Determine the formal charge on each atom in the following molecules. Identify the structure of lower energy in each pair. (a) =-: || | :0: H (b) =c=S (c) H-C=N: :0a: T :0: :8-c=s: | H H-CIN
-
A scatter plot of data is scaled along the y-axis by tens. The data set's residuals are 12, -2, 3, 1.6, -5.4, 3, 0.1, 0.9, and -7. Is the linear model likely strong or weak? How do you know
-
For each account listed, identify whether a debit or credit will cause an increase to that account. Unearned Revenue Answer 1 Choose... Accounts Receivable Answer 2 Choose... Service Revenue Answer 3...
-
Please use Minitab.. If the product feature being measured has a total tolerance of .040" , what is the Gage R&R as a Percent of Engineering Tolerance and as a Percent to Study Variation?...

Study smarter with the SolutionInn App


