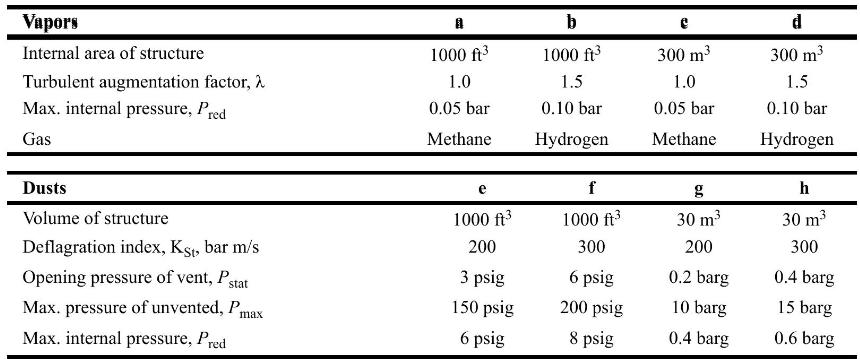
Determine the deflagration vent size for the following structures: Vapors Internal area of structure Turbulent augmentation factor,
Question:
Determine the deflagration vent size for the following structures:
Transcribed Image Text:
Vapors Internal area of structure Turbulent augmentation factor, Max. internal pressure, Pred Gas Dusts Volume of structure Deflagration index, Kst, bar m/s Opening pressure of vent, Pst stat Max. pressure of unvented, P Max. internal pressure, Pred max 8 1000 ft 1.0 0.05 bar Methane e 1000 ft 200 3 psig 150 psig 6 psig b 1000 ft 1.5 0.10 bar Hydrogen f 1000 ft 300 6 psig 200 psig 8 psig 300 m 1.0 0.05 bar Methane 30 m 200 0.2 barg 10 barg 0.4 barg d 300 m 1.5 0.10 bar Hydrogen h 30 m 300 0.4 barg 15 barg 0.6 barg
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
We can utilize the formulae supplied by codes like NFPA 68 which relate the deflagration index Kst t...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
A 10 -ft-wide by \(10-\mathrm{ft}\)-long by \(10-\mathrm{ft}\)-high shed is used to store tanks of methane. What deflagration vent area is required? Assume a maximum internal overpressure of \(0.1...
-
Determine whether the following pairs of structures are actually different compounds or simply resonance forms of the same compounds. (a) (b) (c) (d) (e) (f) (g) (i) (j) and O- and and O- CH C H and...
-
using System; using System.Collections.Generic; using System.ComponentModel; using System.Data; using System.Drawing; using System.Linq; using System.Text; using System.Threading.Tasks; using...
-
For the machine element shown, locate the y coordinate of the center of gravity. 135 i
-
What steps would you take if someone stole your identity?
-
Costsave Corporation, a U'S. company, acquired Denner, a discount supermarket chain in Switzerland, on J anuary 1, 2017. Denner is a subsidiary of Costsave, and its results are consolidated with...
-
Cherokee Company began operations when it issued common stock for $80,000 cash. It paid $60,000 cash in advance for a one-year contract to lease delivery equipment for the business. It signed the...
-
Luke Company has three divisions: Peak, View, and Grand. The company has a hurdle rate of 5.51 percent. Selected operating data for the three divisions follow: Peak View GrandSales revenue $ 33 2...
-
A cooling coil contains ethyl alcohol. The heat capacity of the alcohol is \(0.58 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}\), and its density is \(791 \mathrm{~kg} / \mathrm{m}^{3}\). Determine...
-
Determine the relief diameter for the following two-phase flow conditions. Assume in all cases that \(L / D=0.0\). Reaction mass Volume Set pressure Set temperature (dT/dt)s Maximum pressure Maximum...
-
Write each decimal form in percent form. (a) -0.121 (b) 1.4 (c) 3.2 (d) 4 4
-
Your economics textbook hit the market with a bang, selling 10856 copies in its debut year. The publisher is optimistic, forecasting a sales increase of 11% annually for the next 3 years, followed by...
-
Nike corporation earnings per share in 2005 were $4, and its growth rate during the prior 5 years was 9% per year. If that growth rate were maintained, how long would it take for Nike's earnings per...
-
The Funny-Stay Home (FSH) is an Instagram business. It has been lunched just on February 2020 as a result of the COVID 19 crisis. Mona, the owner of the business needs your help and consultation to...
-
You are proposing that Burger King does a levered recapitalization. Currently, Burger King is financed entirely with equity and has a market capitalization of $1,000,000. Burger King has an inventory...
-
Instructions In a ppt highlight minimum 5 points of topics with the hotel assigned to you- 1. Recruiting 2. Benefits of working with the company 3. Orientation & Training 4. Enlist examples of the...
-
What have the FASB and IASB agreed to do in the Norwalk Agreement?
-
(a) With not more than 300 words, examine why Malaysia missed its 2020 targets to divert 40% of waste from landfills and increase recycling rates to 22%. (10 marks) b) Why arent Malaysian recycling?...
-
A 20.0-mL solution of 0.005 00 M Sn 2+ in 1 M HCl was titrated with 0.020 0 M Ce 4+ to give Sn 4+ and Ce 3+ . Calculate the potential (versus S.C.E.) at the following volumes of Ce 4+ : 0.100, 1.00,...
-
Would indigo tetrasulfonate be a suitable redox indicator for the titration of Fe(CN) 6 4- with Tl 3+ in 1 M HCl? The potential at the equivalence point must be between the potentials for each redox...
-
Compute the titration curve for Demonstration 15-1, in which 400.0 mL of 3.75 mM Fe 2+ are titrated with 20.0 mM MnO 4 - at a fixed pH of 0.00 in 1 M H 2 SO 4 . Calculate the potential versus S.C.E....
-
Please type the computation in an understandable form. Thank you.. Problem 1 You have been given the following information of Lindvie Corporation for September. Actual labor hours used 3,150 hours...
-
1-Company VV uses the weighted-average method in its process costing system. The company has only a single processing department. The company's ending work in process inventory on August 31 consisted...
-
Question 1 - Specialized clerk vs. generalist clerk At a regional life insurance company, the deposit and withdrawal functions associated with a particular investment product are handled by two...

Study smarter with the SolutionInn App


