Estimate the LOC of ethylene using Equations 6-15 and 6-16 in the textbook. Compare to the experimental
Question:
Estimate the LOC of ethylene using Equations 6-15 and 6-16 in the textbook. Compare to the experimental value in Table 6-3.
Table 6-3:
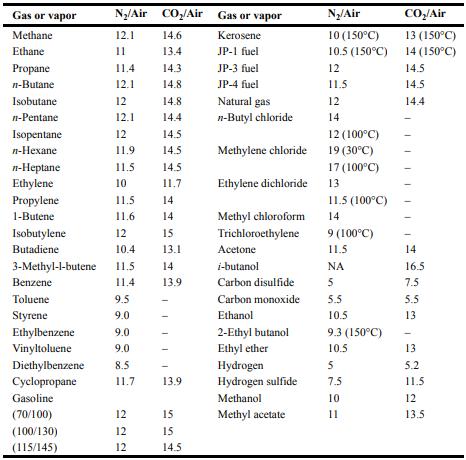
Equation 6-15:
![]()
Equation 6-16:
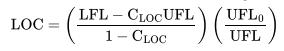
Transcribed Image Text:
Gas or vapor Methane Ethane Propane n-Butane Isobutane n-Pentane Isopentane n-Hexane 12 11.9 11.5 10 11.5 11.6 12 10.4 3-Methyl-l-butene 11.5 11.4 n-Heptane Ethylene Propylene 1-Butene Isobutylene Butadiene Benzene Toluene Styrene Ethylbenzene Vinyltoluene N/Air CO/Air 12.1 14.6 11 13.4 11.4 14.3 12.1 14.8 12 14.8 12.1 14.4 14.5 14.5 Gasoline (70/100) (100/130) (115/145) 9.5 9.0 9.0 9.0 Diethylbenzene 8.5 Cyclopropane 11.7 12 222 12 12 14.5 11.7 14 14 15 13.1 14 13.9 1 13.9 15 15 14.5 Gas or vapor Kerosene JP-1 fuel JP-3 fuel JP-4 fuel Natural gas n-Butyl chloride Methylene chloride Ethylene dichloride Methyl chloroform Trichloroethylene Acetone i-butanol Carbon disulfide Carbon monoxide Ethanol 2-Ethyl butanol Ethyl ether Hydrogen Hydrogen sulfide Methanol Methyl acetate N/Air 10 (150C) 10.5 (150C) 12 11.5 12 14 12 (100C) 19 (30C) 17 (100C) 13 11.5 (100C) 14 9 (100C) 11.5 5 5.5 10.5 9.3 (150C) 10.5 5 7.5 10 11 COy/Air 13 (150C) 14 (150C) 14.5 14.5 14.4 1 14 16.5 7.5 5.5 13 13 5.2 11.5 12 13.5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Equations 615 and 616 can be used to estimate the Limiting Oxygen Concentration LOC of ethylene by u...View the full answer

Answered By

Mercy Kangai
I provide creative and detailed administrative, web search, academic writing, data entry, Personal assistant, Content writing, Translation, Academic writing, editing and proofreading services. I excel at working under tight deadlines with strict expectations. I possess the self-discipline and time management skills necessary to have served as an academic writer for the past seven years. I can bring value to your business and help solve your administrative assistant issues. I have extensive experience in marketing and small business management.
4.80+
27+ Reviews
86+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
Estimate the LOC for (a) carbon monoxide and (b) heptane using Equation 6-15. Compare to experimental values in Table 6-3. Table 6-3: Equation 6-15: Gas or vapor Methane Ethane Propane n-Butane...
-
In a study of hypnotic suggestion, 16 male volunteers were randomly allocated to an experimental group and a control group. Each subject participated in a two-phase experimental session. In the first...
-
To calculate variance and standard deviation, we take the deviations from the mean. At times, we need to consider the deviations from a target value rather than the mean. Consider the case of a...
-
The following scatter plot indicates that 200 150 > 100 50 0 0 20 X 40 O a log x transform may be useful Oa y transform may be useful a x transform may be useful Ono transform is needed Oa 1/x...
-
Determine the volume and the surface area of the chain link shown knowing that it is made from a 0.5-in.-diameter bar and that R = 0.75in and L = 3in.
-
Which of the following are true about Social Security and Medicare tax deductions? A. They are applied to the gross pay. B. Contributions to a 401(k) plan are exempt from these taxes. C. Medicare...
-
There are approximately \(1 \times 10^{12}\) grains of sand in a cubic meter. If the number of grains of sand on the Australian coastline is roughly \(7.5 \times 10^{21}\) grains, roughly how many...
-
The bookkeeper for Lowells Country Music Bar left this incomplete balance sheet. Lowells working capital is $90,000 and its debt to assets ratio is 40 percent. Required Complete the balance sheet by...
-
The XYZ Company just paid a dividend of DO = $1.5 per share, and that dividend is expected to grow at a constant rate of 4.00% for the first 4 years and then 2% per year from year 5 till forever. The...
-
Draw an approximate flammability triangle diagram for methyl alcohol. Use published flammability data from Appendix B and Table 6-3. If a gas containing 20\% methyl alcohol, \(5 \%\) oxygen, and \(75...
-
Estimate the UFL and the LFL for ethylene using the stoichiometric concentrations and Equations 6-10 and 6-11 in the textbook. Compare to the experimental values in Appendix B. Data From Appendix B:...
-
The following are typical questions that might appear on an internal control questionnaire for accounts receivable: 1. Are sales invoices checked for proper pricing, terms, and clerical accuracy? 2....
-
Using Chipotle: Capital structure decision case study Answer the following questions. How does debt policy and the percentage of debt affect Chipotle's firm value Use bullets to highlight the main...
-
Find the local extreme points of f (x) = x e.
-
Case Scenario: Imagine you are a project manager overseeing a team of software developers working on a critical project with a tight deadline. Two team members, Alex and Sarah, have been clashing...
-
Decision Analysis: Myka would like to maintain a 50% gross profit to cover AVS's administrative expenses of US$125,000 and target a 15% net profit. What should the target cost per case be in order to...
-
The restaurant at the Hotel Galaxy offers two choices for breakfast: an all-you-can-eat buffet and an a la carte option, where diners can order from the menu. The buffet option has a budgeted meal...
-
1. Lawrence Company ordered parts costing FC100,000 from a foreign supplier on May 12 when the spot rate was $0.20 per FC. A one-month forward contract was signed on that date to purchase FC100,000...
-
Walker, Inc., is an all-equity firm. The cost of the company's equity is currently 11.4 percent and the risk-free.rate is 3.3 percent. The company is currently considering a project that will cost...
-
The amount of power transmitted by sunlight depends on latitude and the surface area of the solar collector. On a clear day at a certain northern latitude, 0.6 kW/m 2 of solar power strikes the...
-
The property of a fluid called viscosity is related to its internal friction and resistance to being deformed. The viscosity of water, for instance, is less than that of molasses and honey, just as...
-
Referring to the description in Problem P3.16, and given that the viscosity of a certain engine oil is 0.25 kg/(m s), determine the value in the units (a) Poise and (b) Slug/(ft s). Problem P3.16...
-
5. A Firm considers in initiating an aggressive dividend reinvestment plan. In which it allows its investors to use dividends to buy shares at a discount of 40% of current market value. The firms...
-
KC Ltd manufactures and sells a single product. Cost data for the product are given below Variable cost per unit Direct Materials $6 Direct labour $11 Variable manufacturing overhead $5 Variable...
-
. The Yummyr Ice Cream Shoppe sold 9.600 servings of ice cream during June for 55 per serving. The shop purchases the ice cream in large tubs from the Dream Ice Cream Company. Each tub costs the...

Study smarter with the SolutionInn App


