Determine the wavelength of light emitted when an electron in a hydrogen atom makes a transition from
Question:
Determine the wavelength of light emitted when an electron in a hydrogen atom makes a transition from an orbital in n = 6 to an orbital in n = 5.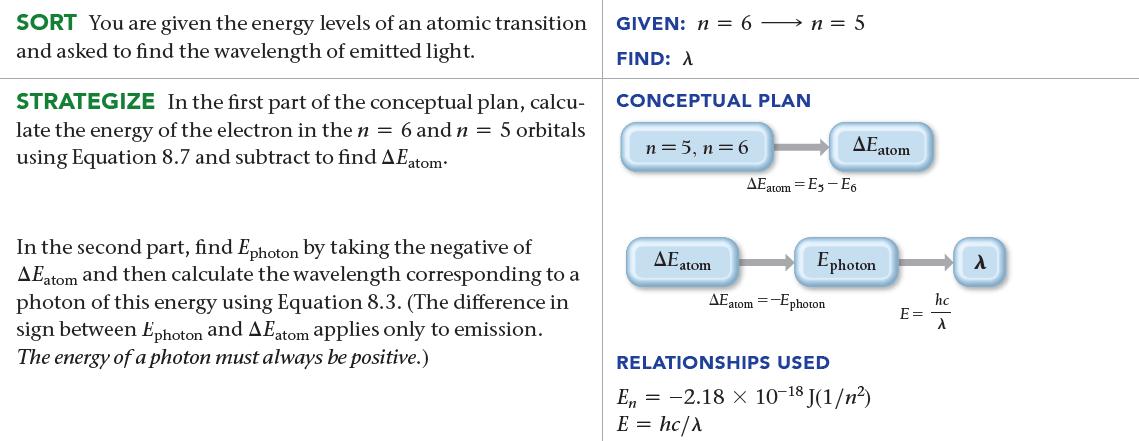
Transcribed Image Text:
SORT You are given the energy levels of an atomic transition and asked to find the wavelength of emitted light. STRATEGIZE In the first part of the conceptual plan, calcu- late the energy of the electron in the n = 6 and n = 5 orbitals using Equation 8.7 and subtract to find AEatom In the second part, find Ephoton by taking the negative of AEatom and then calculate the wavelength corresponding to a photon of this energy using Equation 8.3. (The difference in sign between Ephoton and AEatom applies only to emission. The energy of a photon must always be positive.) GIVEN: n = 6→ n = 5 FIND: A CONCEPTUAL PLAN n = 5, n = 6 AE atom AEatom AE atom-Es-E6 Ephoton AEatom =-Ephoton RELATIONSHIPS USED En = -2.18 × 10-18 J(1/n²) E = hc/λ hc E = - λ λ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
AEatom Es E6 181218 1018 1018 2 218 x 1018 1 5 6 26644 x ...View the full answer

Answered By

RADHIKA MEENAKAR
I am a qualified indian Company Secretary along with Masters in finance with over 6 plus years of professional experience. Apart from this i am a certified accounts and finance tutor on many online platforms.
My Linkedin profile link is here https://www.linkedin.com/in/radhika-meenakar-88b9808a/
5.00+
12+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine the wavelength of light emitted when an electron in a hydrogen atom makes a transition from an orbital in n = 5 to n = 4.
-
Calculate the wavelength of the light emitted when an electron in a hydrogen atom makes each transition and indicate the region of the electromagnetic spectrum (infrared, visible, ultraviolet, etc.)...
-
An electron in a hydrogen atom is excited from the ground state to the n = 4 state. Comment on the correctness of the following statements (true or false). (a) n = 4 is the first excited state. (b)...
-
According to a study conducted by the Gallup organization, the proportion of Americans who are afraid to y is 0.10. A random sample of 1100 Americans results in 121 indicating that they are afraid to...
-
Elbert Company was organized on January 1. During the first year of operations, the following plant asset expenditures and receipts were recorded in random order. Debits 1. Cost of real estate...
-
The rate of return offered by Reliance Insurance Co. on its 20-year annuities is 4.8% compounded monthly. What amount is required to purchase a 20-year annuity with month-end payments of $1000?
-
Which two methods of capital investment analysis ignore present value?
-
Repeat Problem 1 to determine the percentage return on your investment but in this case suppose the price of the stock falls to $7.50 per share. What generalization can be inferred from your answers...
-
reflect on Manufacturing Management's the value chain. What is it? How does it impact success-oriented Manufacturing? Does the issue of good housekeeping impact value? How?
-
On October 1, 2020, Mary Graham organized a computer service company called Echo Systems. Echo is organized as a sole proprietorship and will provide consulting services, computer system...
-
What determines the color of a colored object? Explain why grass appears green.
-
What values of l are possible for n = 3? (a) 0 (or s) (b) 0 and 1 (or s and p) (c) 0, 1, and 2 (or s, p, and d) (d) 0, 1, 2, and 3 (or s, p, d, and f)
-
In Exercises find the indefinite integral. Use a computer algebra system to confirm your result. 1 - sec t cost - 1 dt
-
1) Horizontal Flipping Below example shows how to do horizontal flipping of an image. im imread('cameraman.tif'); data_augmentation is a function thats available to you | im1 = data_augmentation (im,...
-
Use the simplex technique to find the x's which yield the maximum of P, P=2x + 3x + 2 + x3 where x, x2, and x3 are required to be non-negative and to satisfy the constraints that follow: x1 + x2 +...
-
A 45-year-old man was admitted to the hospital for a decompressive laminectomy to treat his long-standing lumbar spinal stenosis. The back pain from the spinal stenosis has not been relieved by pain...
-
Describe 4 principles involved in the decision-making process for Continuous Improvement to enable successful engagement and implementation of improvements
-
Multi objective Problem of Next Investment Advisors1 A client of Next Investment Advisors ("Next") has $80,000 to invest and, as an initial strategy, would like the investment portfolio restricted to...
-
In a simple linear regression analysis, it is found that b1 = 2.556 and s (b1) 4.122. The sample size is n = 22. Conduct an F test for the existence of a linear relationship between the two variables.
-
What is beacon marketing? What are digital wallets?
-
Why can you conclude that the energy of the anti-bonding MO in H + 2 is raised more than the energy of the bonding MO is lowered?
-
The total energy of a molecule is lowered if the orbital energy of the anti-bonding MO is negative, and raised if the orbital energy of the anti-bonding MO is positive. The zero of energy is the...
-
Consider the molecular electrostatic potential map for the LiH molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Construct a probability distribution for the sum shown on the faces when two dices, each with 6 faces, are rolled
-
Introduction External Url 10.1 Two Population Means with Unknown Standard Deviations External Url 10.2 Two Population Means with Known Standard Deviations External Url 10.3 Comparing Two Independent...
-
Select any multinational company and explain the following based on Strategic Outsourcing: 1. Strategic reasons with valid outcomes for outsourcing manufacturing and services. 2. Benefits and...
Contemporary Organizational Behavior From Ideas To Action 1st Edition - ISBN: 0132555883 - Free Book

Study smarter with the SolutionInn App


