Consider a solution of an unknown salt having the general formula BHCl, where B is one of
Question:
Consider a solution of an unknown salt having the general formula BHCl, where B is one of the weak bases in Table 13.3. A 0.10-M solution of the unknown salt has a pH of 5.82. What is the actual formula of the salt?
Table 13.3.
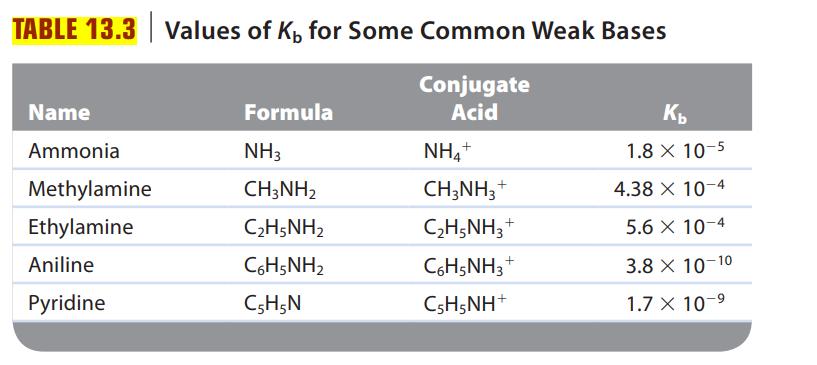
Transcribed Image Text:
TABLE 13.3 Values of K, for Some Common Weak Bases Conjugate Acid Name Ammonia Methylamine Ethylamine Aniline Pyridine Formula NH3 CH3NH₂ C2H5NH2 CoH5NH2 C5H5N NH4+ CH3NH3 + C₂H5NH3 C6H5NH3 CsH5NH* + + Kb 1.8 X 10-5 10-4 10-4 10-10 10-⁹ 4.38 x 5.6 x 3.8 X 1.7 X
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
To determine the actual formula of the unknown salt we need to determine the identity of the weak ba...View the full answer

Answered By

Antony Mutonga
I am a professional educator and writer with exceptional skills in assisting bloggers and other specializations that necessitate a fantastic writer. One of the most significant parts of being the best is that I have provided excellent service to a large number of clients. With my exceptional abilities, I have amassed a large number of references, allowing me to continue working as a respected and admired writer. As a skilled content writer, I am also a reputable IT writer with the necessary talents to turn papers into exceptional results.
4.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
An aqueous solution of an unknown salt of gold is electrolyzed by a current of 2.75 amps for 3.50 hours. The electroplating is carried out with an efficiency of 90.0%, resulting in a deposit of...
-
An aqueous solution of an unknown salt of vanadium is electrolyzed by a current of 2.50 amps for 1.90 hours. The electroplating is carried out with an efficiency of 95.0%, resulting in a deposit of...
-
Separate samples of a solution of an unknown salt are treated with dilute solutions of HBr, H2SO4, and NaOH. A precipitate forms in all three cases. Which of the following cations could the solution...
-
If net assets of a business totalled 200,000 and its total assets on that date amounted to 325,000, its liabilities would amount to: (a) 125,000 (b) 200,000 (c) 525,000 (d) Not sufficient information...
-
Vickie Lynn Smith, an actress and model also known as Anna Nicole Smith, met J. Howard Marshall II in 1991. During their courtship, J. Howard lavished gifts and large sums of money on Anna Nicole,...
-
Refer to the information in QS and instead assume the investment has a salvage value of $20,000. Compute the investments net present value . In QS Following is information on an investment considered...
-
Dominiques Frozen Food Company makes frozen dinners and sells them to retail outlets near London. Dominique has just inherited 10,000 and has decided to invest it in the business. She is trying to...
-
Oceanview School, a private high school, is preparing a planned income statement for the coming academic year ending August 31, 2013. Tuition revenues for the past two years ending August 31 were as...
-
Required information Use the following information for the Exercises below. (Algo) [The following information applies to the questions displayed below.] The following data is provided for Garcon...
-
A. Richard McCarthy (born 2/14/64; Social Security number 100-10-9090) and Christine McCarthy (born 6/1/1966; Social security number 101-21-3434) have a 19-year-old son (born 10/2/99 Social Security...
-
Calculate the pH of each of the following solutions. a. 0.12 M KNO 2 b. 0.45 M NaOCl c. 0.40 M NH 4 ClO 4
-
Are solutions of the following salts acidic, basic, or neutral? For those that are not neutral, write balanced equations for the reactions causing the solution to be acidic or basic. The relevant Ka...
-
Solve. (6x 3 + 7x 2 - 3x) (x 2 - 7) = 0
-
# 34 The Kims deposit $1000 in Wiles Municipal Bond Funds, at 5%. How much is the investment worth (rounded to the nearest cent) at the end of 4 yr, if interest is compounded: a) Annually? The...
-
When a field goal kicker kicks a football as hard as he can at 45 to the horizontal, the ball just clears the 3-m-high crossbar of the goalposts 45.7 m away. (a) What is the maximum speed the kicker...
-
A basketball has a mass of approximately 0.50kg. It carries a uniform charge of 10C. To lift the basketball off the ground, how high above the center of the basketball would you have to hold a 10C...
-
This program checks if a given integer is a prime number. You are to design the processor's FSMD, Datapath and control unit to execute this algorithm. The pseudocode is given below: IsPrime(n): if n
-
After an organization implements its strategic plan, what must management do ?
-
The SAT2010 data file on the text CD contains average reading and math SAT scores for each of the 50 states and Washington D.C. Let the explanatory variable x = reading and the response variable y =...
-
How can NAFTA be beneficial to suppliers of Walmart?
-
Propose an efficient synthesis for the following transformation.
-
For a pair of keto-enol tautomers, explain how IR spectroscopy might be used to identify whether the equilibrium favors the ketone or the enol.
-
Acrolein is an α,β-unsaturated aldehyde that is used in the production of a variety of polymers. Acrolein can be prepared by treating glycerol with an acid catalyst. Propose...
-
Task 2: Response to Exposure Draft "Shaping the Future of Climate-related Accounting and Reporting in Australia" The Australian Accounting Standards Board (AASB) has released Exposure Draft ED SR1...
-
Discussion Topic: Audit Failures The purpose of the audit is to provide assurance as to the accuracy of financial statements. Situations such as the Enron collapse, which were largely due to the...
-
Gastow Pumps is a manufacturer of commercial and heavy industrial Pumps. The firm's two product lines are called Directlift and Gravity. The primary raw materials are flexible steel sheets, and 23cm...

Study smarter with the SolutionInn App


